Nano Res.│郑州大学郭玮/付永柱等:小分子有机硒化物用以调控锂硫电池电化学反应路径
本篇文章版权为郭玮所有,未经授权禁止转载。
背景介绍
锂硫电池被认为是最有潜力的下一代高能量密度电池体系。然而,多硫化物在醚类电解液中的溶解穿梭效应严重阻碍了其实际应用。电解液添加剂被认为是解决锂硫电池穿梭效应、提升电池循环稳定性的一大有效途径。
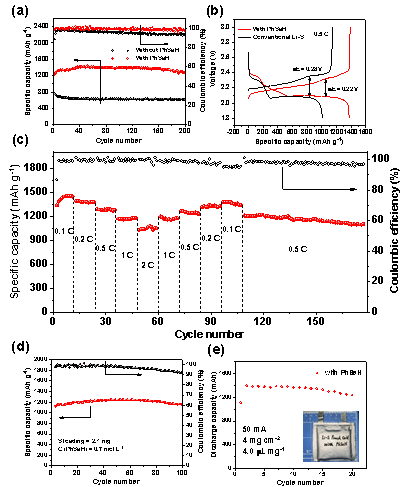
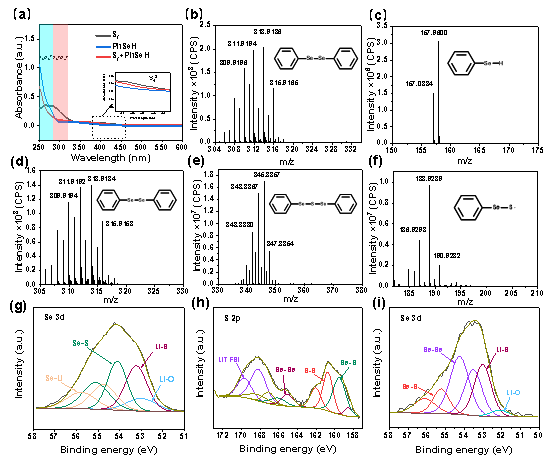
成果简介 本工作报道了一种有机小分子添加剂苯硒酚(PhSeH)应用于锂硫电池。PhSeH能够与硫原位反应生成苯基硒硫化物,从而改变了正极的氧化还原途径,增强了电池的循环稳定性。含有最佳浓度0.70 mol·L−1苯硒酚电解液的锂硫电池,其放电比容量可达1436 mAh·g−1,200圈后容量保持率为92.6%,并展现出相对于传统锂硫电池电解液更低的过电位。在实际应用方面,本工作所报道的含有苯硒酚添加剂的锂硫软包电池总硫含量为1.2 g,E/S低至4.0 L·mg−1,放电容量达到1398mAh,并能保持20圈稳定循环。 图文导读 Figure 1 (a) The optical images and the structure of S8 and PhSeH, (b) long-cycling performance of Li-S cells for 100 cycles, the rate is 0.05 C in the first two cycles and 1 C in the subsequent cycles, and (c) voltage profiles of Li-S cells with the blank and PhSeH electrolytes. Figure 2 (a) The cycling performance and (b) voltage profiles of coin cells with 0.70 M PhSeH and blank electrolytes at the rate of 0.5 C. (c) The rate performance of coin cell with 0.70 M PhSeH electrolyte. (d) Li-S cell with a sulfur mass loading of 2.1 mg and 0.70 M PhSeH electrolyte. (e) The cycling performance of the Li-S pouch cell with PhSeH electrolyte. Figure 3 (a) The UV-Vis spectra of the discharged cathode at cut-off voltage of 2.1 V. (b-c) Mass spectra of the discharged sample. (d-f) Mass spectra of the recharged sample. (g)Se 3d XPS spectra of the discharged sample. (h-i) S 2p (h) and Se 3d (i) XPS spectra of the recharged sample Figure 4 The in-situ Raman spectra of the discharge and charge process on the cathode and the corresponding illustration of the reaction mechanism: (a) the first discharge process and (b) the charge process. 作者简介 文章信息 J. Sun, K. Zhang, Y. Fu, et al. Benzoselenol as an organic electrolyte additive in Li-S battery. Nano Research. https://doi.org/10.1007/s12274-022-4361-z. 识别二维码或点击左下角“阅读原文”可访问全文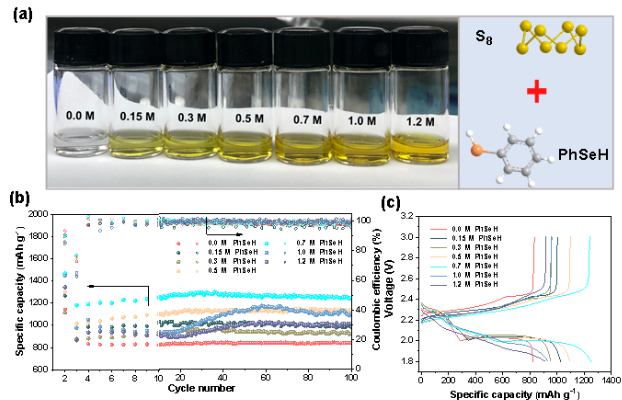



如果篇首注明了授权来源,任何转载需获得来源方的许可!如果篇首未特别注明出处,本文版权属于 X-MOL ( x-mol.com ), 未经许可,谢绝转载!












































 京公网安备 11010802027423号
京公网安备 11010802027423号