Electrocatalysis ( IF 2.7 ) Pub Date : 2019-06-04 , DOI: 10.1007/s12678-019-00538-1 Nnaemeka Nnaji , Njemuwa Nwaji , Gertrude Fomo , John Mack , Tebello Nyokong
Cyclic voltammetry and potentiodynamic polarization techniques were used to study the effects of 4-[4-(1,3-benzothiazol-2yl)phenoxy] phthalonitrile (BT) and tetrakis[(benzo[d]thiazol-2ylphenoxy) phthalocyaninato] gallium(III)chloride (ClGaBTPc) as aluminium corrosion inhibitors in 1.0 M hydrochloric acid. The presence of the inhibitors in the concentration range of 2 to 10 μM was found to retard the aluminium corrosion process such that the inhibition efficiency was found to range from 28.2 to 76.1% for BT and from 71.5 to 82.7% for ClGaBTPc. The latter was a better inhibitor. Scanning electron microscopy and energy-dispersive X-ray measurements reveal effective metal surface protection by the inhibitors, most probably by shielding it from the corrosion attacks of Cl− from the acid. The calculated quantum chemical parameters agreed with experimental results.
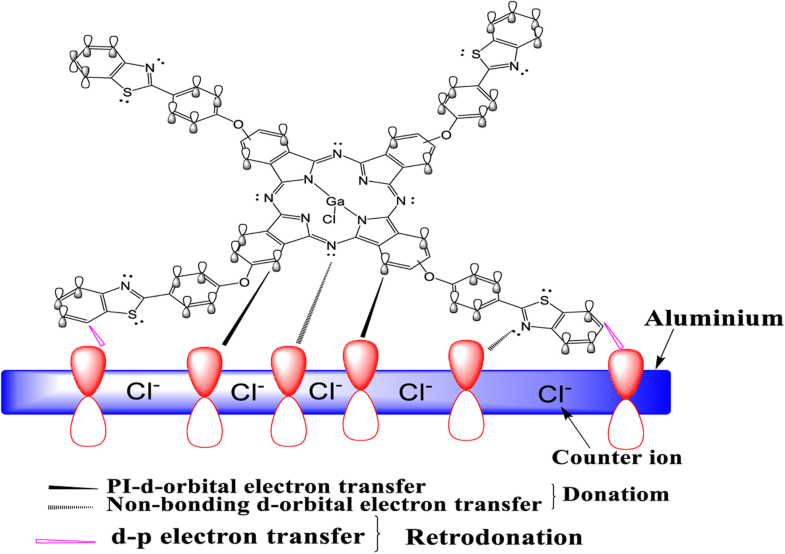
Adsorption of benzothiazole phthalocyanine onto the metal surface for the protection of the metal from the aggressive attack of Cl−.
中文翻译:

苯并噻唑及其酞菁衍生物对铝腐蚀的抑制作用
循环伏安法和电位动力学极化技术用于研究4- [4-(1,3-苯并噻唑-2基)苯氧基]邻苯二甲腈(BT)和四[[苯并[d]噻唑-2基苯氧基]对苯二甲腈]镓(III)的作用氯化物(ClGaBTPc)在1.0 M盐酸中作为铝腐蚀抑制剂。发现浓度在2至10μM范围内的抑制剂可延缓铝腐蚀过程,因此对BT的抑制效率范围为28.2至76.1%,对于ClGaBTPc的抑制效率范围为71.5至82.7%。后者是更好的抑制剂。扫描电子显微镜和能量分散型X射线测量由抑制剂揭示有效金属表面的保护,最可能是由选自Cl的腐蚀攻击屏蔽它-从酸。计算出的量子化学参数与实验结果吻合。

苯并噻唑酞菁在从氯的咄咄逼人的攻势金属保护金属表面的吸附- 。











































 京公网安备 11010802027423号
京公网安备 11010802027423号