RSC主编推荐:有机领域精彩文章快览(免费阅读原文)
英国皇家化学会(RSC)是一个超过175年历史的面向全球化学家的非营利会员制机构,旗下拥有44种期刊,其中很多在化学领域有很高影响力。为了进一步帮助广大读者追踪科技前沿热点,X-MOL团队与英国皇家化学会合作,推出英国皇家化学会期刊主编推荐的精彩文章快览,本期文章属“有机领域”,英文点评来自英国皇家化学会期刊的主编。如果大家对我们的解读有更多的补充和点评,欢迎在文末写评论发表您的高见!
Chemical Science (IF: 9.063)

1. Visible-Light-Mediated Minisci C–H Alkylation of Heteroarenes with Unactivated Alkyl Halides Using O2 as an Oxidant
Chem. Sci., 2019, Advance Article
DOI: 10.1039/C8SC04892D
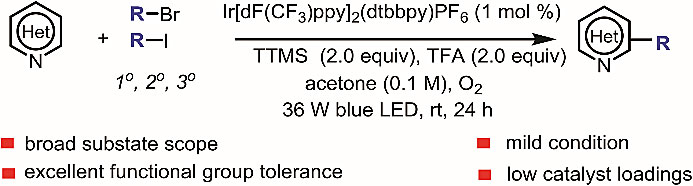
The authors report a method for direct visible-light mediated Minisci C-H alkylation of heteroarenes with unactivated alkyl halides using molecular oxygen as an oxidant at room temperature. They demonstrate the robustness of the method by late-stage functionalisation of complex nitrogen-containing natural products and drugs.
本文报道了一种以分子氧作为氧化剂,室温下可见光介导杂芳香烃和非活化烷基卤化物之间的Minisci C-H键烷基化反应。他们通过复杂含氮天然产物和药物的后期官能化展示了该方法的稳健性。
Open Access(可免费阅读原文)
扫描或长按二维码,识别后直达原文页面,或点此查看原文

2. A Metal-Catalyzed New Approach for α-Alkynylation of Cyclic Amines
Chem. Sci., 2019, Advance Article
DOI: 10.1039/C8SC04115F
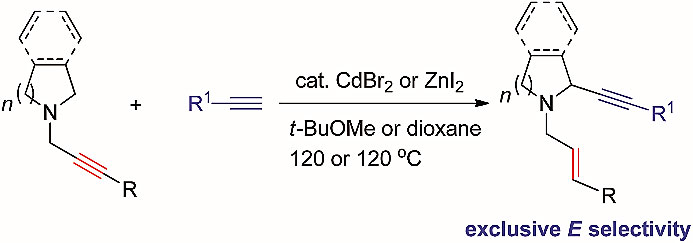
The authors have reported on the first catalytic alpha-alkynylation of cyclic amines utilising the N-propargylic group to prepare 2-(1-alkynyl) N-allylic cyclic amines with an exclusive high E-stereoselectivity for the in situ formed C=C bond. This reaction proceeds via a metal-mediated anti-1,5-hydride transfer forming iminonium intermediate.
本文作者报道了首例环胺的催化α-炔化反应,基于环胺的N-丙炔基团制备了2-(1-炔基)-N-烯丙基环胺,原位形成的C=C键具有高反式立体选择性。反应通过金属介导的反式-1,5-氢迁移形成了亚铵盐中间体。
Open Access(可免费阅读原文)
扫描或长按二维码,识别后直达原文页面,或点此查看原文

Organic Chemistry Frontiers (IF: 5.455)

1. Ni-Catalyzed 1,2-iminoacylation of alkenes via a reductive strategy
Org. Chem. Front., 2018, 5, 3476-3482
DOI: 10.1039/C8QO01044G
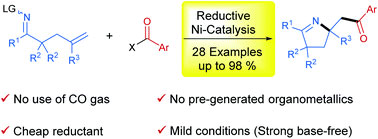
In this protocol, we developed a reductive strategy for 1,2-iminoacylation of alkenes. Under the catalysis of the Ni-biquinoline system, various oxime esters incorporating a pendant terminal olefinic unit were successfully reacted with acid chlorides or anhydrides as electrophilic acylating reagents in the presence of Zn as a reductant, furnishing a series of pyrrolines in moderate to excellent yields. This reaction is distinguished by safe and mild reaction conditions that avoid the use of CO gas as a carbonyl source, pregenerated organometallics and strong bases as reaction additives.
作者发展了一种还原策略实现了烯烃的1,2-亚氨酰化。在Ni/二喹啉体系的催化作用下,以Zn作为还原剂,多种带有侧链端烯烃单元的肟酯与作为亲电酰化试剂的酰基氯或酸酐顺利反应,合成了一系列产率中等至优异的吡咯啉化合物。该反应条件安全且温和,避免了使用一氧化碳气体作为羰基源,也无需使用预先生成的有机金属化合物和强碱作为反应添加剂。
限时免费阅读原文,登陆后可下载
扫描或长按二维码,识别后直达原文页面,或点此查看原文

2. Direct construction of benzimidazo[l,2-c]quinazolin-6-ones via metal-free oxidative C–C bond cleavage
Org. Chem. Front., 2018, Advance Article
DOI: 10.1039/C8QO01039K
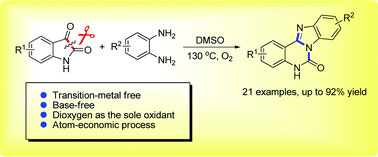
A highly regioselective C–C bond cleavage/amination of isatins has been developed for the synthesis of benzimidazo[1,2-c]quinazolin-6-ones by reacting with o-phenylene diamines. This practical transition-metal-free method is operationally simple, enabling the C–C bond cleavage and triple C–N bond formation, wherein molecular oxygen is the sole required oxidant.
本文介绍了靛红高区域选择性的C-C键裂解/胺化反应,靛红与邻苯二胺反应合成了苯并咪唑[1,2-c]喹唑啉-6-酮。这种无需过渡金属的实用方法操作简单,C-C键发生裂解并形成三个C-N键,反应中分子氧是唯一需要的氧化剂。
限时免费阅读原文,登陆后可下载
扫描或长按二维码,识别后直达原文页面,或点此查看原文

如果篇首注明了授权来源,任何转载需获得来源方的许可!如果篇首未特别注明出处,本文版权属于 X-MOL ( x-mol.com ), 未经许可,谢绝转载!












































 京公网安备 11010802027423号
京公网安备 11010802027423号