PLOS ONE ( IF 2.9 ) Pub Date : 2019-01-16 , DOI: 10.1371/journal.pone.0210548 Jun He , Feng Tang , Daiwen Chen , Bing Yu , Yuheng Luo , Ping Zheng , Xiangbing Mao , Jie Yu , Feng Yu
|
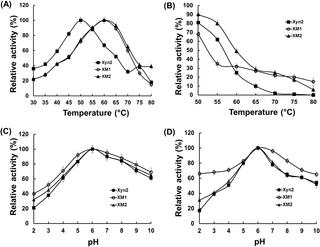
Xylanases isolated from microorganisms such as the Trichoderma reesei have attracted considerable research interest because of their potential in various industrial applications. However, naturally isolated xylanases cannot withstand harsh conditions such as high temperature and basic pH. In this study, we performed structural analysis of the major T. reesei xylanase (Xyn2), and novel flexible regions of the enzyme were identified based on B-factor, a molecular dynamics (MD) parameter. To improve thermostability of the Xyn2, disulfide bonds were introduced into the unstable flexible region by using site-directed mutagenesis and two recombinant xylanases, XM1 (Xyn2Cys12-52) and XM2 (Xyn2Cys59-149) were successfully expressed in Pichia pastoris. Secreted recombinant Xyn2 was estimated by SDS-PAGE to be 24 kDa. Interestingly, the half-lives of XM1 and XM2 at 60°C were 2.5- and 1.8- fold higher, respectively than those of native Xyn2. The XM1 also exhibited improved pH stability and maintained more than 60% activity over pH values ranging from 2.0 to 10.0. However, the specific activity and catalytic efficiency of XM1 was decreased as compared to those of XM2 and native Xyn2. Our results will assist not only in elucidating of the interactions between protein structure and function, but also in rational target selection for improving the thermostability of enzymes.
中文翻译:

里氏木霉的热稳定木聚糖酶的设计,表达和功能表征
从诸如里氏木霉(Trichoderma reesei)之类的微生物中分离出的木聚糖酶由于其在各种工业应用中的潜力而引起了相当大的研究兴趣。然而,天然分离的木聚糖酶不能承受苛刻的条件,例如高温和碱性pH。在这项研究中,我们进行了主要T的结构分析。里氏木聚糖酶(Xyn2),以及基于B因子(一种分子动力学(MD)参数)的酶的新柔性区。为了提高Xyn2的热稳定性,通过定点诱变和两个重组木聚糖酶XM1(Xyn2 Cys12-52)和XM2(Xyn2 Cys59-149)将二硫键引入不稳定的柔性区。)在毕赤酵母中成功表达。通过SDS-PAGE估计分泌的重组Xyn2为24kDa。有趣的是,XM1和XM2在60°C的半衰期分别比天然Xyn2的半衰期高2.5倍和1.8倍。XM1还显示出改善的pH稳定性,并且在2.0到10.0的pH值范围内保持超过60%的活性。但是,与XM2和天然Xyn2相比,XM1的比活和催化效率有所降低。我们的研究结果将不仅有助于阐明蛋白质结构与功能之间的相互作用,而且有助于改善酶的热稳定性的合理靶点选择。











































 京公网安备 11010802027423号
京公网安备 11010802027423号