Journal of Power Sources ( IF 8.1 ) Pub Date : 2018-06-16 , DOI: 10.1016/j.jpowsour.2018.06.051 Krystyna Bachtin , Dominik Kramer , V.S.Kiran Chakravadhanula , Xiaoke Mu , Vanessa Trouillet , Maximilian Kaus , Sylvio Indris , Helmut Ehrenberg , Christina Roth
|
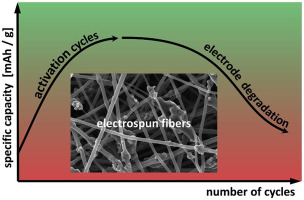
Electrodes prepared by electrospinning often exhibit an activation behavior, i. e. they are reaching their full capacity only after numerous charge and discharge cycles. The activation mechanism can be explained by the improvement of the accessibility of Li+ ions to the active particles of the cathode, which increases with the number of cycles. It is assumed that, as an effect of cycling, the dense, impermeable carbon layer which covers the active material due to the carbonization step during processing cracks and delaminates, allowing this way the Li+ ions to access the active material and to intercalate into it. This has been confirmed by scanning and transmission electron microscopy performed in correlation with the electrochemical performance of electrospun electrodes. However, with even further cycling a decrease in capacity is observed. The microscopic results suggest that this is partly caused by cracks at the carbon-LiFePO4 interfaces. Thus, the cracking responsible for the activation of the electrospun electrodes at the beginning of cycling seems also to cause a part of their degradation at the end of their life. Another slow degradation mechanism confirmed by scanning electron microscopy and by X-ray photoelectron spectroscopy is the ongoing formation of a cathode electrolyte interphase.
中文翻译:

静电纺制的LiFePO 4电池阴极的活化和降解
通过电纺丝制备的电极通常表现出活化行为,即,它们仅在无数次充电和放电循环后才达到其满容量。活化机理可以通过改善Li +离子对阴极活性粒子的可及性来解释,随着循环次数的增加,这种可及性得到提高。假设由于循环的影响,致密的,不可渗透的碳层由于加工过程中的碳化步骤而覆盖了活性材料,从而产生裂纹和分层,从而使Li +离子进入活性物质并嵌入其中。这已经通过与电纺丝电极的电化学性能相关的扫描和透射电子显微镜得到了证实。但是,即使进行进一步的循环,容量也会下降。微观结果表明,这部分是由碳-LiFePO 4界面处的裂纹引起的。因此,在循环开始时负责激活电纺丝电极的开裂似乎也导致其寿命结束时部分降解。扫描电子显微镜和X射线光电子能谱证实的另一种缓慢降解机理是阴极电解质中间相的形成。











































 京公网安备 11010802027423号
京公网安备 11010802027423号