当前位置:
X-MOL 学术
›
Chem. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Direct cyclohexanone oxime synthesis via oxidation-oximization of cyclohexane with ammonium acetate.
Chemical Communications ( IF 4.3 ) Pub Date : 2020-01-08 , DOI: 10.1039/c9cc09840b Ling Peng 1 , Chan Liu 1 , Na Li 1 , Wenzhou Zhong 1 , Liqiu Mao 1 , Steven Robert Kirk 1 , Dulin Yin 1
Chemical Communications ( IF 4.3 ) Pub Date : 2020-01-08 , DOI: 10.1039/c9cc09840b Ling Peng 1 , Chan Liu 1 , Na Li 1 , Wenzhou Zhong 1 , Liqiu Mao 1 , Steven Robert Kirk 1 , Dulin Yin 1
Affiliation
|
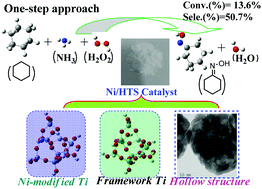
An unexpected cascade reaction for oxidation-oximization of cyclohexane with ammonium acetate was developed for the first time to access cyclohexanone oxime with 50.7% selectivity (13.6% conversion). Tetrahedral Ti sites in Ni-containing hollow titanium silicalite can serve as bifunctional catalytic centers in the reaction. This methodology not only provides a direct approach to prepare cyclohexanone oxime, but also simplifies process chemistry. Various available nitrogen sources, such as ammonium salt and even ammonia can be used as starting materials.
中文翻译:

通过用乙酸铵对环己烷进行氧化-肟化来直接合成环己酮肟。
首次开发了出乎意料的级联反应,用乙酸铵将环己烷氧化-肟化,获得选择性为50.7%(转化率为13.6%)的环己酮肟。含镍的中空钛硅质岩中的四面体Ti位点可作为反应中的双功能催化中心。该方法不仅提供了制备环己酮肟的直接方法,而且简化了过程化学。各种可用的氮源,例如铵盐,甚至氨水,都可以用作起始原料。
更新日期:2020-01-08
中文翻译:

通过用乙酸铵对环己烷进行氧化-肟化来直接合成环己酮肟。
首次开发了出乎意料的级联反应,用乙酸铵将环己烷氧化-肟化,获得选择性为50.7%(转化率为13.6%)的环己酮肟。含镍的中空钛硅质岩中的四面体Ti位点可作为反应中的双功能催化中心。该方法不仅提供了制备环己酮肟的直接方法,而且简化了过程化学。各种可用的氮源,例如铵盐,甚至氨水,都可以用作起始原料。











































 京公网安备 11010802027423号
京公网安备 11010802027423号