当前位置:
X-MOL 学术
›
J. Heterocycl. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Diastereoselective synthesis of pyrrolo[1, 2‐c]imidazoles using chiral thiohydantoins, malononitrile, and aldehydes and evaluation of their antioxidant and antibacterial activities
Journal of Heterocyclic Chemistry ( IF 2.0 ) Pub Date : 2019-12-20 , DOI: 10.1002/jhet.3762 Khadijeh Mollanejad 1 , Sakineh Asghari 1, 2 , Khosrow Jadidi 3
Journal of Heterocyclic Chemistry ( IF 2.0 ) Pub Date : 2019-12-20 , DOI: 10.1002/jhet.3762 Khadijeh Mollanejad 1 , Sakineh Asghari 1, 2 , Khosrow Jadidi 3
Affiliation
|
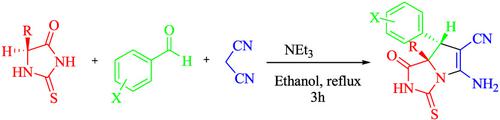
Diastereoselective synthesis of pyrrolo[1,2‐c]imidazoles is reported from three‐component reaction of chiral thiohydantoins, aldehydes, and malononitrile in the presence of NEt3. The antioxidant properties of the obtained products were evaluated by 1,1‐diphenyl‐2‐picrylhydrazyl (DPPH) radical scavenging assay. Among the products, compound 4l possessing NH2 and NH groups and bromine atom at 4‐position of the aromatic ring displayed the highest antioxidant activity (90%). Also, their antibacterial activities were explored against gram‐positive and gram‐negative bacteria using disc diffusion method. Among the synthesized compounds, 4a with chlorine atom at para position of the aromatic ring, and methyl group (the smallest alkyl group) at 7‐position, displayed the best antibacterial activity against the tested gram‐positive bacteria.
中文翻译:

使用手性硫代乙内酰脲,丙二腈和醛的非对映选择性合成吡咯并[1,2–c]咪唑并评估其抗氧化和抗菌活性
据报道,在NEt 3存在下,手性硫代乙内酰脲,醛类和丙二腈的三组分反应可实现对吡咯并[1,2-c]咪唑的非对映选择性合成。通过1,1-二苯基-2-吡啶并肼基(DPPH)自由基清除试验评估了所得产品的抗氧化性能。在这些产品中,具有NH 2和NH基团的化合物4l和芳环4位的溴原子表现出最高的抗氧化活性(90%)。此外,使用圆盘扩散法探索了它们对革兰氏阳性和革兰氏阴性细菌的抗菌活性。在合成的化合物中,4a 芳环对位的氯原子和7位的甲基(最小的烷基)对被测革兰氏阳性菌表现出最佳的抗菌活性。
更新日期:2019-12-21
中文翻译:

使用手性硫代乙内酰脲,丙二腈和醛的非对映选择性合成吡咯并[1,2–c]咪唑并评估其抗氧化和抗菌活性
据报道,在NEt 3存在下,手性硫代乙内酰脲,醛类和丙二腈的三组分反应可实现对吡咯并[1,2-c]咪唑的非对映选择性合成。通过1,1-二苯基-2-吡啶并肼基(DPPH)自由基清除试验评估了所得产品的抗氧化性能。在这些产品中,具有NH 2和NH基团的化合物4l和芳环4位的溴原子表现出最高的抗氧化活性(90%)。此外,使用圆盘扩散法探索了它们对革兰氏阳性和革兰氏阴性细菌的抗菌活性。在合成的化合物中,4a 芳环对位的氯原子和7位的甲基(最小的烷基)对被测革兰氏阳性菌表现出最佳的抗菌活性。







































 京公网安备 11010802027423号
京公网安备 11010802027423号