当前位置:
X-MOL 学术
›
Catal. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Josiphos-type binaphane ligands for the asymmetric Ir-catalyzed hydrogenation of acyclic aromatic N-aryl imines
Catalysis Communications ( IF 3.4 ) Pub Date : 2019-12-10 , DOI: 10.1016/j.catcom.2019.105906 Dongxu Zhang , Xiaowei Zhou , Ruixia Liu , Muqiong Li , Xiaoye Li , Ru Jiang , Huifang Nie , Shengyong Zhang
中文翻译:

Josiphos型二苯甲醚配体用于不对称的Ir催化的无环芳族N-芳基亚胺的加氢反应
更新日期:2019-12-11
Catalysis Communications ( IF 3.4 ) Pub Date : 2019-12-10 , DOI: 10.1016/j.catcom.2019.105906 Dongxu Zhang , Xiaowei Zhou , Ruixia Liu , Muqiong Li , Xiaoye Li , Ru Jiang , Huifang Nie , Shengyong Zhang
|
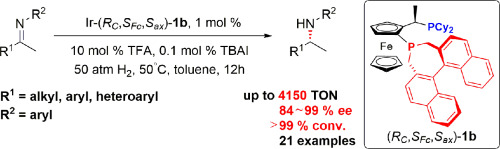
The Ir-catalyzed asymmetric hydrogenation of acyclic aromatic N-aryl imines with Josiphos-type binaphane ligands has been described. Under the optimized conditions, a wide range of imines were hydrogenated to afford the corresponding chiral amines in high yields (up to 94%) and good to excellent enantioselectivities (up to >99% ee). The synthetic utility of the present protocol has been demonstrated through the asymmetric synthesis of the key chiral intermediates of calcium sensing receptor modulators on a gram scale.
中文翻译:

Josiphos型二苯甲醚配体用于不对称的Ir催化的无环芳族N-芳基亚胺的加氢反应
已经描述了具有Josiphos型联苯甲酰胺配体的无环芳族N-芳基亚胺的Ir催化的不对称氢化。在优化的条件下,将广泛的亚胺氢化,以高收率(高达94%)和良好至优异的对映选择性(高达> 99%ee)提供相应的手性胺。本协议的合成效用已通过以克为单位的钙敏感受体调节剂的关键手性中间体的不对称合成得到了证明。











































 京公网安备 11010802027423号
京公网安备 11010802027423号