Structure ( IF 4.4 ) Pub Date : 2023-04-13 , DOI: 10.1016/j.str.2023.03.012 Sarah Bonhomme 1 , Carlos Contreras-Martel 1 , Andréa Dessen 1 , Pauline Macheboeuf 1
|
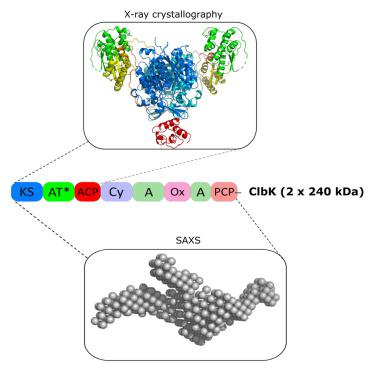
The genotoxin colibactin produced by Escherichia coli is involved in the development of colorectal cancers. This secondary metabolite is synthesized by a multi-protein machinery, mainly composed of non-ribosomal peptide synthetase (NRPS)/polyketide synthase (PKS) enzymes. In order to decipher the function of a PKS-NRPS hybrid enzyme implicated in a key step of colibactin biosynthesis, we conducted an extensive structural characterization of the ClbK megaenzyme. Here we present the crystal structure of the complete trans-AT PKS module of ClbK showing structural specificities of hybrid enzymes. In addition, we report the SAXS solution structure of the full-length ClbK hybrid that reveals a dimeric organization as well as several catalytic chambers. These results provide a structural framework for the transfer of a colibactin precursor through a PKS-NRPS hybrid enzyme and can pave the way for re-engineering PKS-NRPS hybrid megaenzymes to generate diverse metabolites with many applications.
中文翻译:

参与基因毒素大肠菌素生物合成的 PKS-NRPS 杂合巨酶的结构
大肠杆菌产生的基因毒素大肠菌素与结直肠癌的发展有关。这种次级代谢物由多蛋白机制合成,主要由非核糖体肽合成酶 (NRPS)/聚酮化合物合成酶 (PKS) 组成。为了破译涉及大肠菌素生物合成关键步骤的 PKS-NRPS 杂合酶的功能,我们对 ClbK 巨型酶进行了广泛的结构表征。在这里,我们展示了完整反式的晶体结构-ClbK 的 AT PKS 模块显示出杂合酶的结构特异性。此外,我们报告了全长 ClbK 杂种的 SAXS 溶液结构,该结构揭示了二聚体组织以及多个催化室。这些结果为大肠菌素前体通过 PKS-NRPS 杂合酶的转移提供了结构框架,并可为重新设计 PKS-NRPS 杂合巨酶以生成具有多种应用的多种代谢物铺平道路。











































 京公网安备 11010802027423号
京公网安备 11010802027423号