European Journal of Medicinal Chemistry ( IF 6.0 ) Pub Date : 2022-11-24 , DOI: 10.1016/j.ejmech.2022.114949 Liliya F Saifina 1 , Mohnad Abdalla 2 , Liliya M Gubaidullina 1 , Irina V Zueva 1 , Wafa Ali Eltayb 3 , Amr Ahmed El-Arabey 4 , Alexandra D Kharlamova 1 , Oksana A Lenina 1 , Vyacheslav E Semenov 1 , Konstantin A Petrov 5
|
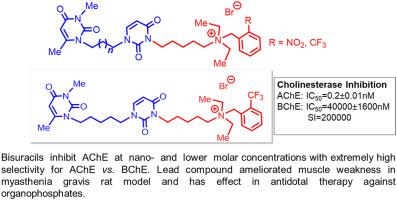
A series of new compounds in which uracil and 3,6-dimethyluracil moieties are bridged with different spacers were prepared and evaluated in vitro for the acetyl- and butyrylcholinesterase (AChE and BChE) inhibitory activities. These bisuracils are shown to be very effective inhibitors of AChE, inhibiting the enzyme at nano- and lower molar concentrations with extremely high selectivity for AChE vs. BChE. Kinetic analysis showed that the lead compound 2h acts as a slow-binding inhibitor of AChE and possess a long drug-target residence time (τ = 1/koff = 18.6 ± 7.5 min). Moreover, compound 2h ameliorated muscle weakness in myasthenia gravis rat model with a lower effective dose and longer lasting effect than pyridostigmine bromide. Besides, it was shown that compound 2h has an effect of increasing efficiency of antidotal therapy as a pretreatment for poisoning by organophosphates.
中文翻译:

基于尿嘧啶部分的新型慢结合可逆乙酰胆碱酯酶抑制剂可能用于治疗重症肌无力和防止有机磷中毒
制备了一系列新化合物,其中尿嘧啶和 3,6-二甲基尿嘧啶部分与不同的间隔物桥接,并在体外评估了乙酰胆碱酯酶和丁酰胆碱酯酶(AChE 和 BChE)的抑制活性。这些双尿嘧啶被证明是非常有效的 AChE 抑制剂,在纳米级和更低的摩尔浓度下抑制酶,对 AChE相对于BChE具有极高的选择性。动力学分析表明,先导化合物2h作为 AChE 的慢结合抑制剂,具有较长的药物靶点停留时间(τ = 1/k off = 18.6 ± 7.5 分钟)。此外,化合物2h改善重症肌无力大鼠模型的肌肉无力,有效剂量低于溴化吡啶斯的明且持续时间更长。此外,显示化合物2h作为有机磷中毒的预处理具有提高解毒治疗效率的作用。











































 京公网安备 11010802027423号
京公网安备 11010802027423号