当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Directed, Remote Dirhodium C(sp3)-H Functionalization, Desaturative Annulation, and Desaturation
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2022-09-26 , DOI: 10.1021/jacs.2c07427 Sailu Munnuri 1 , John R Falck 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2022-09-26 , DOI: 10.1021/jacs.2c07427 Sailu Munnuri 1 , John R Falck 1
Affiliation
|
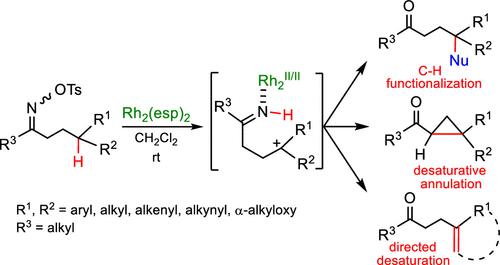
Iminodirhodium reactive intermediates generated in situ from O-tosyloximes using Rh2(esp)2 in CH2Cl2 at rt were exploited for an agile trichotomy of challenging transformations: (1) remote C–H functionalizations using an exceptionally broad diversity of inorganic and organic nucleophiles including several unconventional examples, for example, ethers and acyl silanes; (2) desaturative annulation, a biomimetic 1,3-methylene C–C ring-closure with an overall loss of two hydrogens; and (3) directed desaturation for the acceptor-less, regioselective creation of γ,δ- or γ,δ,ε,ζ-olefins. Compared with typical iminyl transition-metal-mediated and 1,5-hydrogen atom-transfer (1,5-HAT) processes, iminodirhodium intermediates are largely underexplored, especially with respect to C(sp3)-H centers and, yet, have the potential to be transformative by virtue of their substrate breadth, regiocontrol, and elusive reaction modality. A substrate scope includes benzylic, allylic, propargylic, tertiary, and α-alkyloxy centers.
中文翻译:

定向、远程 Dirhodium C(sp3)-H 功能化、去饱和环化和去饱和
在 rt 使用CH 2 Cl 2中的Rh 2 (esp) 2从O - tosyloximes原位生成的亚氨基二铑反应中间体被用于具有挑战性的转化的灵活三分法:(1)使用极其广泛的无机和有机亲核试剂,包括几个非常规的例子,例如醚和酰基硅烷;(2)去饱和环化,一个仿生 1,3-亚甲基 C-C 闭环,总体损失了两个氢;(3) 定向去饱和,用于无受体、区域选择性地生成 γ,δ-或 γ,δ,ε,ζ-烯烃。与典型的亚氨基过渡金属介导和 1,5-氢原子转移 (1,5-HAT) 过程相比,亚氨基二铑中间体在很大程度上尚未得到充分探索,尤其是在 C(sp 3 )-H 中心方面,然而,凭借其底物广度、区域控制和难以捉摸的反应方式而具有变革性的潜力。底物范围包括苄基、烯丙基、炔丙基、叔基和 α-烷氧基中心。
更新日期:2022-09-26
中文翻译:

定向、远程 Dirhodium C(sp3)-H 功能化、去饱和环化和去饱和
在 rt 使用CH 2 Cl 2中的Rh 2 (esp) 2从O - tosyloximes原位生成的亚氨基二铑反应中间体被用于具有挑战性的转化的灵活三分法:(1)使用极其广泛的无机和有机亲核试剂,包括几个非常规的例子,例如醚和酰基硅烷;(2)去饱和环化,一个仿生 1,3-亚甲基 C-C 闭环,总体损失了两个氢;(3) 定向去饱和,用于无受体、区域选择性地生成 γ,δ-或 γ,δ,ε,ζ-烯烃。与典型的亚氨基过渡金属介导和 1,5-氢原子转移 (1,5-HAT) 过程相比,亚氨基二铑中间体在很大程度上尚未得到充分探索,尤其是在 C(sp 3 )-H 中心方面,然而,凭借其底物广度、区域控制和难以捉摸的反应方式而具有变革性的潜力。底物范围包括苄基、烯丙基、炔丙基、叔基和 α-烷氧基中心。











































 京公网安备 11010802027423号
京公网安备 11010802027423号