Journal of Hepatology ( IF 26.8 ) Pub Date : 2022-09-20 , DOI: 10.1016/j.jhep.2022.09.006 Masatoshi Kudo , Richard S. Finn , Shukui Qin , Kwang-Hyub Han , Kenji Ikeda , Ann-Lii Cheng , Arndt Vogel , Francesco Tovoli , Kazuomi Ueshima , Hiroshi Aikata , Carlos López López , Marc Pracht , Zhiqiang Meng , Bruno Daniele , Joong-Won Park , Daniel Palmer , Toshiyuki Tamai , Kenichi Saito , Corina E. Dutcus , Riccardo Lencioni
|
Background & Aims
Validated surrogate endpoints for overall survival (OS) are important for expediting the clinical study and drug-development processes. Herein, we aimed to validate objective response as an independent predictor of OS in individuals with unresectable hepatocellular carcinoma (HCC) receiving systemic anti-angiogenic therapy.
Methods
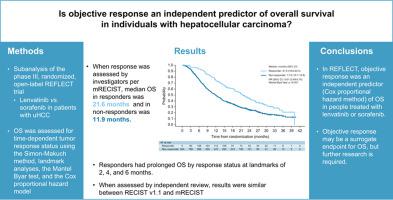
We investigated the association between objective response (investigator-assessed mRECIST, independent radiologic review [IRR] mRECIST and RECIST v1.1) and OS in REFLECT, a phase III study of lenvatinib vs. sorafenib. We conducted landmark analyses (Simon-Makuch) of OS by objective response at 2, 4, and 6 months after randomization.
Results
Median OS was 21.6 months (95% CI 18.6–24.5) for responders (investigator-assessed mRECIST) vs. 11.9 months (95% CI 10.7–12.8) for non-responders (hazard ratio [HR] 0.61; 95% CI 0.49–0.76; p <0.001). Objective response by IRR per mRECIST and RECIST v1.1 supported the association with OS (HR 0.61; 95% CI 0.51–0.72; p <0.001 and HR 0.50; 95% CI 0.39–0.65; p <0.001, respectively). OS was significantly prolonged for responders vs. non-responders (investigator-assessed mRECIST) at the 2-month (HR 0.61; 95% CI 0.49–0.76; p <0.001), 4-month (HR 0.63; 95% CI 0.51–0.80; p <0.001), and 6-month (HR 0.68; 95% CI 0.54–0.86; p <0.001) landmarks. Results were similar when assessed by IRR, with both mRECIST and RECIST v1.1. An exploratory multivariate Cox regression analysis identified objective response by investigator-assessed mRECIST (HR 0.55; 95% CI 0.44–0.68; p <0.0001) and IRR-assessed RECIST v1.1 (HR 0.49; 95% CI, 0.38–0.64; p <0.0001) as independent predictors of OS in individuals with unresectable HCC.
Conclusions
Objective response was an independent predictor of OS in individuals with unresectable HCC in REFLECT; additional studies are needed to confirm surrogacy. Participants achieving a complete or partial response by mRECIST or RECIST v1.1 had significantly longer survival vs. those with stable/progressive/non-evaluable disease.
ClinicalTrials.gov number
NCT01761266.
Impact and implications
This analysis of data taken from a completed clinical trial (REFLECT) looked for any link between objective response and overall survival time in individuals with unresectable HCC receiving anti-angiogenic treatments. Significantly longer median overall survival was found for responders (21.6 months) vs. non-responders (11.9 months). Overall survival was also significantly longer for responders vs. non-responders (based on objective response status at 2, 4, and 6 months) in the landmark analysis. Our results indicate that objective response is an independent predictor of overall survival in this setting, confirming its validity as a rapid marker of efficacy that can be applied in phase II trials; however, further validation is required to determine is validity for other systemic treatments (e.g. immunotherapies), or as a surrogate of overall survival.
中文翻译:

晚期不可切除肝细胞癌的总生存期和客观反应:REFLECT 研究的亚组分析
背景与目标
经过验证的总生存期 (OS) 替代终点对于加快临床研究和药物开发过程非常重要。在此,我们旨在验证客观反应作为接受全身抗血管生成治疗的无法切除的肝细胞癌 (HCC) 患者 OS 的独立预测因子。
方法
我们在 REFLECT 中调查了客观反应(研究者评估的 mRECIST、独立放射学审查 [IRR] mRECIST 和 RECIST v1.1)与 OS 之间的关联,REFLECT 是乐伐替尼与索拉非尼的 III 期研究。我们在随机分组后 2、4 和 6 个月通过客观反应对 OS 进行了里程碑式分析 (Simon-Makuch)。
结果
应答者(研究者评估的 mRECIST)的中位 OS 为 21.6 个月(95% CI 18.6–24.5),而无应答者为11.9 个月(95% CI 10.7–12.8)(风险比 [HR] 0.61;95% CI 0.49– 0.76;p < 0.001)。根据 mRECIST 和 RECIST v1.1 的 IRR 的客观反应支持与 OS 的关联(分别为 HR 0.61;95% CI 0.51–0.72;p < 0.001 和 HR 0.50;95% CI 0.39–0.65;p < 0.001)。在 2 个月(HR 0.61;95% CI 0.49–0.76 ;p < 0.001)、4 个月(HR 0.63;95% CI 0.51– 0.80;p < 0.001)和 6 个月(HR 0.68;95% CI 0.54–0.86;p <0.001) 地标。通过 IRR 评估时,mRECIST 和 RECIST v1.1 的结果相似。探索性多变量 Cox 回归分析通过研究者评估的 mRECIST(HR 0.55;95% CI 0.44–0.68;p < 0.0001)和 IRR 评估的 RECIST v1.1(HR 0.49;95% CI,0.38–0.64;p <0.0001) 作为不可切除 HCC 患者 OS 的独立预测因子。
结论
在 REFLECT 中,客观反应是不可切除 HCC 患者 OS 的独立预测因子;需要更多的研究来确认代孕。通过 mRECIST 或 RECIST v1.1 获得完全或部分反应的参与者与患有稳定/进展/不可评估疾病的参与者相比,生存期明显更长。
ClinicalTrials.gov 编号
NCT01761266。
影响和启示
这项对来自已完成临床试验 (REFLECT) 的数据的分析旨在寻找接受抗血管生成治疗的无法切除的 HCC 患者的客观反应与总生存时间之间的任何联系。发现有反应者(21.6 个月)与. 无反应者(11.9 个月)。应答者与应答者的总生存期也明显更长。界标分析中的无反应者(基于 2、4 和 6 个月的客观反应状态)。我们的结果表明,客观反应是这种情况下总生存期的独立预测因子,证实了其作为可应用于 II 期试验的快速疗效标志物的有效性;然而,需要进一步验证以确定是否适用于其他全身治疗(例如免疫疗法),或作为总生存期的替代指标。











































 京公网安备 11010802027423号
京公网安备 11010802027423号