Water Research ( IF 11.4 ) Pub Date : 2022-09-16 , DOI: 10.1016/j.watres.2022.119126 Jingli Ren 1 , Yutong Liu 1 , Weimin Cao 1 , Liyang Zhang 1 , Fen Xu 2 , Juan Liu 3 , Yubo Wen 4 , Jian Xiao 5 , Lei Wang 6 , Xiaoxiong Zhuo 7 , Junfeng Ji 1 , Yuanyuan Liu 1
|
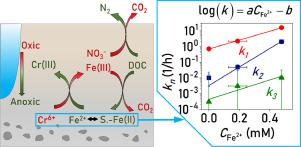
Sediment-associated Fe(II) is a critical reductant for immobilizing groundwater contaminants, such as Cr(VI). The reduction reactivity of sediment-associated Fe(II) is dependent on its binding environment and influenced by the biogeochemical transformation of other elements (i.e., C, N and Mn), challenging the description and prediction of the reactivity of Fe(II) in natural sediments. Here, anaerobic batch experiments were conducted to study the variation in sediment-associated Fe(II) reactivity toward Cr(VI) in natural sediments collected from an intensive agricultural area located in Guangxi, China, where nitrate is a common surface water and groundwater contaminant. Then, a process-based model was developed to describe the coupled biogeochemical processes of C, N, Mn, Fe, and Cr. In the process-based model, Cr(VI) reduction by sediment-associated Fe(II) was described using a previously developed multirate model, which categorized the reactive Fe(II) into three fractions based on their extractabilities in sodium acetate and HCl solutions. The experimental results showed that Fe(II) generation was inhibited by NO3- and/or NO2−. After NO3− and NO2− were exhausted, the Fe(II) content and its reduction rate toward Cr(VI) increased rapidly. As the Fe(II) content increased, the three reactive Fe(II) fractions exhibited approximately linear correlations with aqueous Fe(II) concentrations (), which was probably driven by sorptive equilibrium and redox equilibrium between aqueous and solid phases. The model results indicated that the reaction rate constants of the three Fe(II) fractions (kn) significantly increased with incubation time, and log(kn) correlated well with [, and ]. The numerical model developed in this study provides an applicable method to describe and predict Cr(VI) removal from groundwater under dynamic redox conditions.











































 京公网安备 11010802027423号
京公网安备 11010802027423号