Applied Catalysis B: Environment and Energy ( IF 20.2 ) Pub Date : 2022-08-18 , DOI: 10.1016/j.apcatb.2022.121862 Kaifeng Wang , Ran Mao , Rui Liu , Juanjuan Zhang , Xu Zhao
|
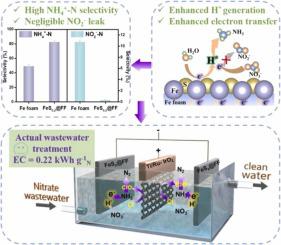
The electrocatalytic reduction of nitrate (NO3−) to ammonia (NH4+) and further oxidation to N2 via active chlorine is an effective method for denitrification. High activity, NH4+–N selectivity and long-term stability of a cathode are essential for the application. Herein, we reported a sulfur-modified Fe foam (FeSx @FF) cathode, which achieved high NO3−–N conversion (94.96%) and NH4+–N selectivity (82.29%) due to its enhanced electron transfer, atomic hydrogen (H*) generation, and adsorption of NO3−. Combined in-situ differential electrochemical mass spectrometry and theoretical calculations reveal that the ultrafast *NO3 reduction to *NO2 and low formation energy of *NO are crucial in reduction of *NO3 to *NH3. The FeS0.2 @FF cathode for realistic industrial wastewater (∼350 mg L−1 NO3−–N) treatment shows high TN removal (90.75%) and N2 selectivity (98.88%) with an ultralow electrical cost (∼0.22 kWh g−1N). This designed system has potential for efficient NO3− removal in the saline wastewater.
中文翻译:

硫掺杂促进的自支撑铁阴极电催化还原硝酸盐:选择性、稳定性和潜在机理
将硝酸盐(NO 3 -)电催化还原为氨(NH 4 +)并通过活性氯进一步氧化为N 2是一种有效的反硝化方法。正极的高活性、NH 4 + -N 选择性和长期稳定性对于该应用至关重要。在此,我们报道了一种硫改性的泡沫铁 (FeS x @FF )阴极,由于其增强的电子转移、原子氢气 (H*) 的产生和 NO 3的吸附-. 结合原位差分电化学质谱和理论计算表明,*NO 3超快还原为*NO 2和*NO的低形成能是*NO 3还原为*NH 3的关键。用于实际工业废水(~350 mg L -1 NO 3 - -N)处理的 FeS 0.2 @FF 阴极显示出高 TN 去除率(90.75%)和 N 2选择性(98.88%)以及超低的电力成本(~0.22 kWh g -1 N)。这种设计的系统具有在含盐废水中有效去除NO 3 -的潜力。











































 京公网安备 11010802027423号
京公网安备 11010802027423号