Water Research ( IF 11.4 ) Pub Date : 2022-08-14 , DOI: 10.1016/j.watres.2022.118981 Jianbing Wang 1 , Jiyong Liu 2 , Xianjia Peng 3 , Mengchang He 4 , Xingyun Hu 5 , Jinmin Zhao 3 , Feng Zhu 3 , Xin Yang 3 , Linghao Kong 5
|
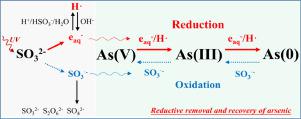
The removal of arsenic (As(V) and As(III)) from contaminated water has attracted great attention. However, the generation of arsenic-containing hazardous waste by traditional methods has become an inevitable environmental problem. Herein, a UV/sulfite advanced reduction method was proposed to remove As(V) and As(III) from aqueous solution in the form of valuable elemental arsenic (As(0)), thus avoiding the generation of arsenic-containing hazardous waste. The results showed that greater than 99.9% of As(V) and As(III) were reduced to the high purity As(0) (> 99.5 wt%) with the residual arsenic concentration below 10 μg L−1. The hydrated electrons (eaq−), H• and SO3•− radicals are generated by the UV/sulfite process, of which eaq− and H• serve as reductants of As(V) and As(III) while the SO3•− radicals inhibit arsenic reduction by oxidizing arsenic. The effective quantum efficiency (Φ) for the formation of As(0) in the As(V) and As(III) removal process is approximately 0.0078 and 0.0055 mol/Einstein, respectively. The reduction of arsenic is favorable under alkaline conditions (pH > 9.0) due to the higher photolysis efficiency of SO32− than HSO3− (pKa = 7.2) and higher stability of eaq−/H• under alkaline conditions. The presence of dissolved oxygen (O2), NO2−, NO3−, CO32−, PO43− and humic acid (HA) inhibited arsenic reduction through light blocking or eaq−/H• scavenging effects while Cl−, SO42−, Ca2+ and Mg2+ had negligible effects on arsenic reduction. The proposed method can effectively remove and recover arsenic from contaminated water at a low cost, demonstrating feasibility for practical application. This study provides a novel technology for the reductive removal and recovery of arsenic from contaminated water.
中文翻译:

紫外/亚硫酸盐法从水溶液中还原去除 As(V) 和 As(III):元素砷的回收
从污染水中去除砷(As(V)和As(III))引起了人们的极大关注。然而,传统方法产生含砷危险废物已成为不可避免的环境问题。在此,提出了一种紫外/亚硫酸盐高级还原方法,从水溶液中以有价值的元素砷 (As(0)) 的形式去除 As(V) 和 As(III),从而避免了含砷危险废物的产生。结果表明,99.9% 以上的 As(V) 和 As(III) 被还原为高纯度 As(0) (> 99.5 wt%),残留砷浓度低于 10 μg L -1。水合电子(e aq -)、H •和 SO 3 •-UV/亚硫酸盐过程产生自由基,其中e aq -和H •作为As(V) 和As(III) 的还原剂,而SO 3 •-自由基通过氧化砷抑制砷还原。在 As(V) 和 As(III) 去除过程中形成 As(0) 的有效量子效率 (Φ) 分别约为 0.0078 和 0.0055 mol/Einstein。由于 SO 3 2-的光解效率高于 HSO 3 - (pK a = 7.2) 和 e aq - /H •在碱性条件下。溶解氧 (O 2 )、NO 2 -、NO 3 -、CO 3 2-、PO 4 3-和腐植酸 (HA) 的存在通过阻光或 e aq - /H •清除作用抑制砷还原,而 Cl - , SO 4 2- , Ca 2+和 Mg 2+对砷还原的影响可以忽略不计。所提出的方法可以以低成本有效地从污染水中去除和回收砷,具有实际应用的可行性。该研究为从污染水中还原去除和回收砷提供了一种新技术。







































 京公网安备 11010802027423号
京公网安备 11010802027423号