Talanta ( IF 6.1 ) Pub Date : 2022-08-02 , DOI: 10.1016/j.talanta.2022.123789 Wenyan Li 1 , Xingzhuo Zhou 2 , Wenjun Yan 3 , Ruihan Wang 2 , Zeqian Yang 2 , Yongchong Hu 2 , Yang Liu 2 , Zhichao Jia 2 , Yingqi Li 4
|
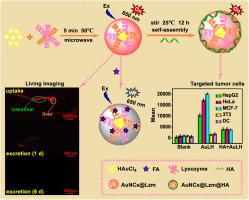
Herein, lysozyme-encapsulated gold nanoclusters (AuNCs@Lzm, AuL) were favorably obtained by a simple and economical synthesis. The resulting AuL exhibits low toxicity, exceptional stability and intense red fluorescence at 650 nm, which can enter living cells and is mainly located in lysosomes. The AuL selectively and sensitively drove to detect folic acid (FA) with a detection limit as low as 0.19 nM based on the combination of the static quenching and the internal filtering effect. Interestingly, the fascinating results discovered that the AuL with ignorable toxicity was adsorbed from the intestine into the liver, and essentially was cleared from the body in 6 days without significant bioaccumulation in zebrafish. Furthermore, the hyaluronic acid (HA) coating AuLH exhibits remarkably targeted tumors towards MCF-7>HeLa > HepG2»NIH/3T3≈DC, which attributed to the number of receptors expressed by the cells. All these advantages highlight that the AuL is a versatile nanoplatform for sensing, in vivo fluorescence imaging and tumor targeting.
中文翻译:

溶菌酶包裹的金纳米团簇用于叶酸的超灵敏检测和体内成像
在此,溶菌酶包裹的金纳米簇(AuNCs@Lzm,AuL)通过简单且经济的合成有利地获得。所得的 AuL 具有低毒性、出色的稳定性和 650 nm 处的强烈红色荧光,可进入活细胞,主要位于溶酶体中。基于静态猝灭和内部过滤效应的结合,AuL选择性和灵敏地检测叶酸(FA),检测限低至0.19 nM。有趣的是,令人着迷的结果发现,毒性可忽略不计的 AuL 从肠道吸附到肝脏,并在 6 天内基本上从体内清除,而在斑马鱼中没有明显的生物蓄积。此外,透明质酸 (HA) 涂层 AuLH 表现出对 MCF-7>HeLa > HepG2»NIH/3T3≈DC 的显着靶向肿瘤,这归因于细胞表达的受体数量。所有这些优势都凸显了 AuL 是一种用于传感的多功能纳米平台,体内荧光成像和肿瘤靶向。



























 京公网安备 11010802027423号
京公网安备 11010802027423号