Chemical Engineering Science ( IF 4.1 ) Pub Date : 2022-07-22 , DOI: 10.1016/j.ces.2022.117936 Zhiwen Wang , Huan Wang , Yingzhang Shi , Cheng Liu , Ling Wu , Shijing Liang
|
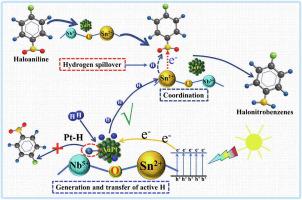
AuPt alloy/SnNb2O6 ultrathin nanosheet (AuPt/SN) as a photocatalyst is constructed for selective hydrogenation of p-chloronitrobenzene (p-CNB) under visible light irradiation. A typical catalyst (Au0.5Pt0.5/SN) exhibits a high conversion of halonitrobenzenes (99.8%) and selectivity of haloaniline (99.1%). The results of in situ ATR-IR, XPS and Raman indicate a preferential chemoselective adsorption of –NO2 that Lewis base sites (Sn2+) on the catalyst could selectively coordinate with –NO2 whereas C-Cl bond is not be bonded, improving the catalytic selectivity. Atom Pt in alloy is responsible for the formation of ·H, while Au inhabits the dissociation of C-Cl via repressing the generation of Pt-H. The photogenerated electrons can accelerate the ·H formation. ·H could be transferred to Sn2+ sites for the hydrogenation of –NO2 by hydrogen spillover. Therefore, the high performance of the catalyst can be attributed to a surface synergetic effect among Lewis base sites (Sn2+), AuPt alloy and photogenerated electrons.
中文翻译:

表面工程改进对氯硝基苯在可见光下AuPt合金/SnNb2O6超薄纳米片上的选择性加氢
构建了AuPt合金/SnNb 2 O 6超薄纳米片(AuPt/SN)作为光催化剂,用于在可见光照射下选择性加氢对氯硝基苯(p -CNB)。典型的催化剂(Au 0.5 Pt 0.5 /SN)表现出高的卤代硝基苯转化率(99.8%)和卤代苯胺的选择性(99.1%)。原位 ATR-IR、XPS 和拉曼的结果表明催化剂上的路易斯碱基 (Sn 2+ ) 可以选择性地与 -NO 2配位 -NO 2的优先化学选择性吸附而C-Cl键不键合,提高了催化选择性。合金中的原子Pt负责·H的形成,而Au通过抑制Pt-H的生成来抑制C-Cl的解离。光生电子可以加速·H的形成。·H可以通过氢溢出转移到Sn 2+位点用于-NO 2加氢。因此,催化剂的高性能可归因于路易斯碱基(Sn 2+)、AuPt合金和光生电子之间的表面协同效应。











































 京公网安备 11010802027423号
京公网安备 11010802027423号