Molecular Therapy ( IF 12.1 ) Pub Date : 2022-07-12 , DOI: 10.1016/j.ymthe.2022.07.006 Zoe Raglow 1 , Mary Kathryn McKenna 2 , Challice L Bonifant 3 , Wenjing Wang 4 , Marina Pasca di Magliano 5 , Johannes Stadlmann 6 , Josef M Penninger 7 , Richard D Cummings 8 , Malcolm K Brenner 9 , David M Markovitz 10
|
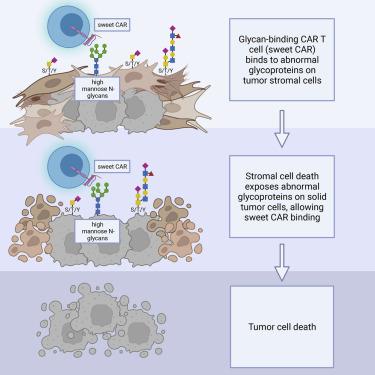
Chimeric antigen receptor (CAR) T cell therapy has created a paradigm shift in the treatment of hematologic malignancies but has not been as effective toward solid tumors. For such tumors, the primary obstacles facing CAR T cells are scarcity of tumor-specific antigens and the hostile and complex tumor microenvironment. Glycosylation, the process by which sugars are post-translationally added to proteins or lipids, is profoundly dysregulated in cancer. Abnormally glycosylated glycoproteins expressed on cancer cells offer unique targets for CAR T therapy as they are specific to tumor cells. Tumor stromal cells also express abnormal glycoproteins and thus also have the potential to be targeted by glycan-binding CAR T cells. This review will discuss the state of CAR T cells in the therapy of solid tumors, the cancer glycoproteome and its potential for use as a therapeutic target, and the landscape and future of glycan-binding CAR T cell therapy.
中文翻译:

CAR 疗法的靶向聚糖:甜 CAR 的出现
嵌合抗原受体 (CAR) T 细胞疗法在血液恶性肿瘤的治疗中创造了范式转变,但对实体瘤的治疗效果不佳。对于此类肿瘤,CAR T细胞面临的主要障碍是肿瘤特异性抗原的缺乏以及恶劣而复杂的肿瘤微环境。糖基化是糖在翻译后添加到蛋白质或脂质中的过程,在癌症中严重失调。癌细胞上表达的异常糖基化糖蛋白为 CAR T 疗法提供了独特的靶点,因为它们对肿瘤细胞具有特异性。肿瘤基质细胞也表达异常糖蛋白,因此也有可能被聚糖结合 CAR T 细胞靶向。这篇综述将讨论 CAR T 细胞在实体瘤治疗中的现状、癌症糖蛋白组及其作为治疗靶点的潜力,以及聚糖结合 CAR T 细胞治疗的前景和未来。











































 京公网安备 11010802027423号
京公网安备 11010802027423号