当前位置:
X-MOL 学术
›
J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Nickel-Catalyzed Tandem Ueno–Stork Cyclization: Stereoselective 1,2-Dicarbofunctionalization of Cyclic Alkenes
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2022-06-22 , DOI: 10.1021/acs.joc.2c00770 Pedro de Andrade Horn 1 , Hunter S Sims 1 , Mingji Dai 1
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2022-06-22 , DOI: 10.1021/acs.joc.2c00770 Pedro de Andrade Horn 1 , Hunter S Sims 1 , Mingji Dai 1
Affiliation
|
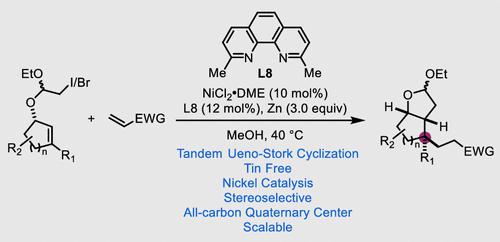
A nickel-catalyzed tandem Ueno–Stork cyclization is developed to enable stereoselective 1,2-dicarbofunctionalization of cyclic alkenes and efficiently build various bicyclic products. This new protocol does not involve any toxic or difficult-to-remove tin reagent and is scalable and amenable to build all-carbon quaternary centers.
中文翻译:

镍催化串联 Ueno-Stork 环化:环状烯烃的立体选择性 1,2-二碳官能化
开发了一种镍催化的串联 Ueno-Stork 环化,以实现环状烯烃的立体选择性 1,2-二碳官能化,并有效地构建各种双环产物。这个新协议不涉及任何有毒或难以去除的锡试剂,并且具有可扩展性并且可以构建全碳四元中心。
更新日期:2022-06-22
中文翻译:

镍催化串联 Ueno-Stork 环化:环状烯烃的立体选择性 1,2-二碳官能化
开发了一种镍催化的串联 Ueno-Stork 环化,以实现环状烯烃的立体选择性 1,2-二碳官能化,并有效地构建各种双环产物。这个新协议不涉及任何有毒或难以去除的锡试剂,并且具有可扩展性并且可以构建全碳四元中心。











































 京公网安备 11010802027423号
京公网安备 11010802027423号