Environment International ( IF 10.3 ) Pub Date : 2022-06-15 , DOI: 10.1016/j.envint.2022.107351 Qiyu Wang 1 , Jing Huang 2 , Shuai Liu 1 , Caiyun Wang 3 , Yuanxiang Jin 3 , Hong Lai 1 , Wenqing Tu 4
|
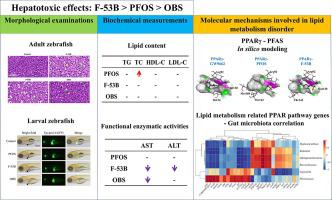
Perfluorooctane sulfonate (PFOS) has been reported to induce hepatotoxicity in wildlife and humans. Novel PFOS alternatives have been widely used following restrictions on PFOS, but little is known about their potential toxicity. Here, the first comprehensive investigation on the chronic hepatotoxicity and underlying molecular mechanisms of PFOS, 6:2Cl-PFESA (F-53B), and sodium p-perfluorous nonenoxybenzene sulfonate (OBS) was carried out on adult zebrafish through a histopathological examination, biochemical measurement, and multi-omics analysis. PFOS and its alternatives caused changes in liver histopathology and liver function indices in the order of F-53B > PFOS > OBS, which was consistent with their concentration in the liver. In silico modeling and transcriptional profiles suggested that the aberrant hepatic lipid metabolism induced by F-53B and PFOS was initiated by the action on peroxisome proliferator-activated receptor γ (PPARγ), which triggered changes in downstream genes transcription and led to an imbalance between lipid synthesis and expenditure. Gut microbiome analysis provided another novel mechanistic perspective that changes in the abundance of Legionella, Ralstonia, Brevundimonas, Alphaproteobacteria, Plesiomonas, and Hyphomicrobium might link to alterations in the PPAR pathway based on their significant correlation. This study provides insight into the molecular mechanisms of hepatotoxicity induced by PFOS and its novel alternatives and highlights the need for concern about their environmental exposure risks.
中文翻译:

与肠道微生物群失调相关的肝脏脂质代谢异常会引发成年斑马鱼新型全氟辛烷磺酸替代品的肝毒性
据报道,全氟辛烷磺酸 (PFOS) 会引起野生动物和人类的肝毒性。在对 PFOS 进行限制后,新型 PFOS 替代品已被广泛使用,但对其潜在毒性知之甚少。在这里,通过组织病理学检查、生化检查,对成年斑马鱼进行了PFOS、6:2Cl-PFESA (F-53B) 和对全氟壬氧基苯磺酸钠 (OBS ) 的慢性肝毒性和潜在分子机制的首次全面研究。测量和多组学分析。PFOS及其替代品引起肝脏组织病理学和肝功能指标的变化顺序为F-53B>PFOS>OBS,这与其在肝脏中的浓度一致。计算机模型和转录谱表明,F-53B 和 PFOS 诱导的异常肝脏脂质代谢是由过氧化物酶体增殖物激活受体 γ (PPARγ) 的作用引发的,这引发了下游基因转录的变化,导致脂质之间的不平衡。综合和支出。肠道微生物组分析提供了另一种新颖的机制视角,即军团菌、拉尔斯顿氏菌、短波单胞菌、α变形菌、邻单胞菌和丝微菌的丰度变化可能与 PPAR 途径的改变有关,因为它们具有显着的相关性。这项研究深入了解了 PFOS 及其新型替代品引起肝毒性的分子机制,并强调了关注其环境暴露风险的必要性。











































 京公网安备 11010802027423号
京公网安备 11010802027423号