Structure ( IF 4.4 ) Pub Date : 2022-04-15 , DOI: 10.1016/j.str.2022.03.013 Franziska Jäger 1 , Anaïs Lamy 2 , Wei-Sheng Sun 2 , Nina Guerini 1 , Ronnie P-A Berntsson 2
|
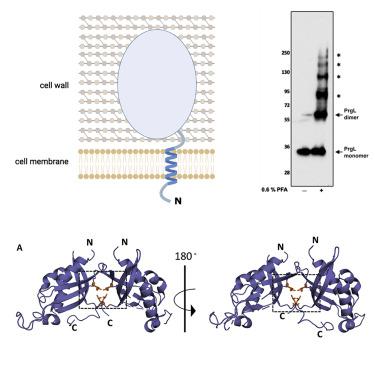
Multidrug-resistant bacteria pose serious problems in hospital-acquired infections (HAIs). Most antibiotic resistance genes are acquired via conjugative gene transfer, mediated by type 4 secretion systems (T4SS). Although most multidrug-resistant bacteria responsible for HAIs are of Gram-positive origin, with enterococci being major contributors, mostly Gram-negative T4SSs have been characterized. Here, we describe the structure and organization of PrgL, a core protein of the T4SS channel, encoded by the pCF10 plasmid from Enterococcus faecalis. The structure of PrgL displays similarity to VirB8 proteins of Gram-negative T4SSs. In vitro experiments show that the soluble domain alone is enough to drive both dimerization and dodecamerization, with a dimerization interface that differs from all other known VirB8-like proteins. In vivo experiments verify the importance of PrgL dimerization. Our findings provide insight into the molecular building blocks of Gram-positive T4SS, highlighting similarities but also unique features in PrgL compared to other VirB8-like proteins.
中文翻译:

肠球菌 T4SS 蛋白 PrgL 的结构揭示了 VirB8 蛋白家族中独特的二聚化界面
耐多药细菌在医院获得性感染 (HAI) 中造成严重问题。大多数抗生素抗性基因是通过结合基因转移获得的,由 4 型分泌系统 (T4SS) 介导。尽管导致 HAI 的大多数耐多药细菌是革兰氏阳性菌,肠球菌是主要贡献者,但大多数革兰氏阴性 T4SS 已被表征。在这里,我们描述了 PrgL 的结构和组织,它是 T4SS 通道的核心蛋白,由粪肠球菌的 pCF10 质粒编码。PrgL 的结构与革兰氏阴性 T4SS 的 VirB8 蛋白相似。体外实验表明,单独的可溶结构域足以驱动二聚化和十二聚化,二聚化界面不同于所有其他已知的 VirB8 样蛋白。体内实验验证了 PrgL 二聚化的重要性。我们的研究结果提供了对革兰氏阳性 T4SS 分子构建模块的深入了解,突出了 PrgL 与其他 VirB8 样蛋白相比的相似性和独特性。











































 京公网安备 11010802027423号
京公网安备 11010802027423号