Applied Catalysis B: Environment and Energy ( IF 20.2 ) Pub Date : 2021-11-15 , DOI: 10.1016/j.apcatb.2021.120914 Shuang Yang 1 , Ji-Yu Zhu 1 , Xiao-Nan Chen 1 , Meng-Jie Huang 1 , Sheng-Hao Cai 1 , Ji-Yuan Han 1 , Ji-Sen Li 1
|
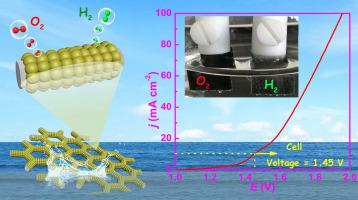
Rational design and exploitation of efficient and inexpensive catalysts for water electrolysis are highly desired, yet very challenging. Herein, for the first time, we report a nanostructured catalyst of nickel phosphides-ruthenium phosphides self-supported on nickel foam (Ni2P-Ru2P/NF) through an in situ growth-phosphorization process. As expected, by virtue of prominent intrinsic activity, rich electrochemically active sites, and high electronic conductivity, the resultant Ni2P-Ru2P/NF exhibits enhanced electrocatalytic behavior for the oxygen evolution reaction and hydrogen evolution reaction, which delivers low overpotentials of 160 and 101 mV at 10 mA cm 2 in alkaline media, respectively. Remarkably, the Ni2P-Ru2P/NF can dramatically accelerate full water splitting with an ultralow cell voltage of 1.45 V at 10 mA cm−2, which far exceeds the benchmark Pt-C/NF//RuO2/NF (1.64 V) and ranks among the best electrocatalysts previously reported.
2 in alkaline media, respectively. Remarkably, the Ni2P-Ru2P/NF can dramatically accelerate full water splitting with an ultralow cell voltage of 1.45 V at 10 mA cm−2, which far exceeds the benchmark Pt-C/NF//RuO2/NF (1.64 V) and ranks among the best electrocatalysts previously reported.
中文翻译:

用于增强电化学水分解的具有人工异质界面的自支撑双金属磷化物
非常需要合理设计和开发高效且廉价的水电解催化剂,但也非常具有挑战性。在此,我们首次通过原位生长磷化工艺报道了一种自担载在泡沫镍上的磷化镍-磷化钌纳米结构催化剂(Ni 2 P-Ru 2 P/NF)。正如预期的那样,由于显着的本征活性、丰富的电化学活性位点和高电子电导率,所得的 Ni 2 P-Ru 2 P/NF 对析氧反应和析氢反应表现出增强的电催化行为,从而提供了较低的过电位160 和 101 mV 在 10 mA cm  2分别在碱性介质中。值得注意的是,Ni 2 P-Ru 2 P/NF 可以在 10 mA cm -2 下以 1.45 V 的超低电池电压显着加速全水分解,远远超过基准 Pt-C/NF//RuO 2 /NF( 1.64 V)并且是先前报道的最好的电催化剂之一。
2分别在碱性介质中。值得注意的是,Ni 2 P-Ru 2 P/NF 可以在 10 mA cm -2 下以 1.45 V 的超低电池电压显着加速全水分解,远远超过基准 Pt-C/NF//RuO 2 /NF( 1.64 V)并且是先前报道的最好的电催化剂之一。











































 京公网安备 11010802027423号
京公网安备 11010802027423号