Applied Catalysis B: Environment and Energy ( IF 20.2 ) Pub Date : 2021-11-02 , DOI: 10.1016/j.apcatb.2021.120877 Xiaoying Liang 1 , Di Wang 1 , Zhiyu Zhao 1 , Tong Li 1 , Zhenhuan Chen 1 , Yaowen Gao 1 , Chun Hu 1
|
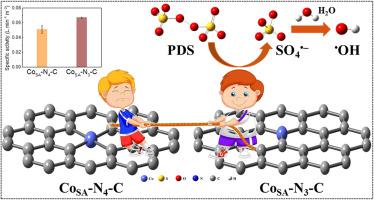
Two atomically dispersed cobalt catalysts with different nitrogen coordination numbers (denoted as CoSA-Nx-C) were synthesized and firstly compared to activate peroxydisulfate (PDS) for bisphenol A (BPA) degradation. Theoretical calculations unveiled that lowering the Co N coordination number from four to three can apparently increase the electron density of the single Co atom in CoSA-N3-C to enhance PDS conversion. The low-coordinated CoSA-N3-C with Co
N coordination number from four to three can apparently increase the electron density of the single Co atom in CoSA-N3-C to enhance PDS conversion. The low-coordinated CoSA-N3-C with Co N3 coordination structure displays a high specific activity of 0.067 L min
N3 coordination structure displays a high specific activity of 0.067 L min 1 m
1 m 2, which is 1.31 times greater than that of CoSA-N4-C with normal Co
2, which is 1.31 times greater than that of CoSA-N4-C with normal Co N4 configuration (0.051 L min
N4 configuration (0.051 L min 1 m
1 m 2) in PDS activation. Electron paramagnetic resonance (EPR) measurements and quenching tests confirmed the primary role of sulfate radical (SO4•−) in BPA oxidation over CoSA-N3-C with PDS. Moreover, CoSA-N3-C delivers favorable durability for PDS activation and potential practicability for realistic wastewater remediation. These findings provide a novel and useful avenue to coordination number modulation of SACs for wider environmental applications.
2) in PDS activation. Electron paramagnetic resonance (EPR) measurements and quenching tests confirmed the primary role of sulfate radical (SO4•−) in BPA oxidation over CoSA-N3-C with PDS. Moreover, CoSA-N3-C delivers favorable durability for PDS activation and potential practicability for realistic wastewater remediation. These findings provide a novel and useful avenue to coordination number modulation of SACs for wider environmental applications.
中文翻译:

设计低配位单钴原子以促进过硫酸盐活化以增强有机污染物氧化
合成了两种具有不同氮配位数的原子分散钴催化剂(表示为 Co SA -N x -C),并首先比较活化过二硫酸盐 (PDS) 对双酚 A (BPA) 的降解。理论计算表明,将 Co  N 配位数从 4降低到 3 可以明显增加 Co SA -N 3 -C 中单个 Co 原子的电子密度,从而增强 PDS 转换。具有Co N 3配位结构的低配位Co SA -N 3 -C显示出0.067 L min 1 m 2的高比活度,是Co的1.31倍
N 配位数从 4降低到 3 可以明显增加 Co SA -N 3 -C 中单个 Co 原子的电子密度,从而增强 PDS 转换。具有Co N 3配位结构的低配位Co SA -N 3 -C显示出0.067 L min 1 m 2的高比活度,是Co的1.31倍

 SA -N 4 -C在 PDS 活化中具有正常的 Co
SA -N 4 -C在 PDS 活化中具有正常的 Co  N 4配置(0.051 L min
N 4配置(0.051 L min  1 m
1 m  2)。电子顺磁共振 (EPR) 测量和猝灭测试证实了硫酸根 (SO 4 •- ) 在使用 PDS 的Co SA -N 3 -C 上的BPA 氧化中的主要作用。此外,Co SA -N 3 -C 为 PDS 活化提供了良好的耐久性和现实废水修复的潜在实用性。这些发现为 SAC 的配位数调制提供了一种新颖且有用的途径,以用于更广泛的环境应用。
2)。电子顺磁共振 (EPR) 测量和猝灭测试证实了硫酸根 (SO 4 •- ) 在使用 PDS 的Co SA -N 3 -C 上的BPA 氧化中的主要作用。此外,Co SA -N 3 -C 为 PDS 活化提供了良好的耐久性和现实废水修复的潜在实用性。这些发现为 SAC 的配位数调制提供了一种新颖且有用的途径,以用于更广泛的环境应用。











































 京公网安备 11010802027423号
京公网安备 11010802027423号