当前位置:
X-MOL 学术
›
Eur. J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
C–H Amination Enabled [2+1+1+1] Annulation Reaction in Water: Access to Benzoxazoles
European Journal of Organic Chemistry ( IF 2.5 ) Pub Date : 2021-09-30 , DOI: 10.1002/ejoc.202100942 Huanfeng Jiang 1 , Fulin Chen 2 , Chuanle Zhu 2 , Zhiyi Yang 2 , Chi Liu 2 , Hao Zeng 2 , Wanqing Wu 2
European Journal of Organic Chemistry ( IF 2.5 ) Pub Date : 2021-09-30 , DOI: 10.1002/ejoc.202100942 Huanfeng Jiang 1 , Fulin Chen 2 , Chuanle Zhu 2 , Zhiyi Yang 2 , Chi Liu 2 , Hao Zeng 2 , Wanqing Wu 2
Affiliation
|
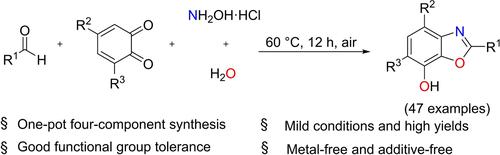
A novel and efficient strategy to prepare benzoxazoles through a [2+1+1+1] annulation of benzo-1,2-quinones, aldehydes, hydroxylamine hydrochloride and water is reported. The method is operationally simple and gram-scalable; it has a broad substrate scope and valuable functional group tolerance, and ensures high yields.

中文翻译:

C–H 胺化使水中的 [2+1+1+1] 环化反应:获得苯并恶唑
报道了一种通过苯并-1,2-醌、醛、盐酸羟胺和水的 [2+1+1+1] 环化制备苯并恶唑的新型有效策略。该方法操作简单且可扩展;它具有广泛的底物范围和宝贵的官能团耐受性,并确保高产量。

更新日期:2021-11-23

中文翻译:

C–H 胺化使水中的 [2+1+1+1] 环化反应:获得苯并恶唑
报道了一种通过苯并-1,2-醌、醛、盐酸羟胺和水的 [2+1+1+1] 环化制备苯并恶唑的新型有效策略。该方法操作简单且可扩展;它具有广泛的底物范围和宝贵的官能团耐受性,并确保高产量。












































 京公网安备 11010802027423号
京公网安备 11010802027423号