Chem Catalysis ( IF 11.5 ) Pub Date : 2021-09-23 , DOI: 10.1016/j.checat.2021.08.014 Haibo Yin 1 , Zhen Chen 1 , Shangchao Xiong 1 , Jianjun Chen 1 , Chizhong Wang 1 , Rong Wang 1 , Yasutaka Kuwahara 2 , Jingshan Luo 3 , Hiromi Yamashita 2 , Yue Peng 1 , Junhua Li 1
|
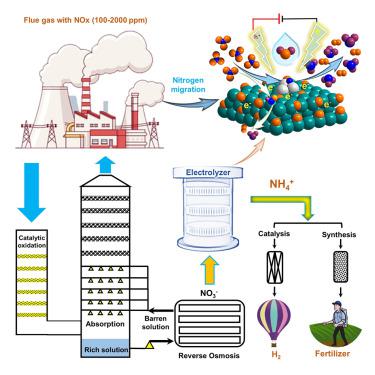
Electrocatalytic conversion of nitrate (NO3−) to ammonia (NH3) holds significant potential in the control of nitrogen oxide (NOx) from stationary sources. However, previous studies on reaction intermediates remain unclear. Here we report that PdCu/Cu2O hybrids with mesoporous hollow sphere structure show high selectivity (96.70%) and Faradaic efficiency (94.32%) for NH3 synthesis from NO3−. Detailed characterizations demonstrate that (1) Pd enables electron transfer (Pd 3d → Cu 3d) and causes the polarization of Cu 3d orbitals by forming partial PdCu alloys, which makes Pd electron deficient but offers empty orbits to adsorb NO3−, and (2) electron-rich Cu is more conducive to the occurrence of NO3− reduction. The mutual confirmation of online differential electrochemical mass spectrometry and density functional theory calculations demonstrates that PdCu alloys block the generation of ∗NOH intermediate and facilitate the formation of ∗N, providing a new mechanism for NH3 synthesis from NO3− reduction reactions.
中文翻译:

合金效应诱导的电子极化驱动硝酸盐电还原为氨
硝酸盐 (NO 3 - ) 向氨 (NH 3 ) 的电催化转化在控制来自固定源的氮氧化物 (NO x ) 方面具有巨大潜力。然而,之前对反应中间体的研究仍不清楚。在这里,我们报告了具有介孔空心球结构的PdCu/Cu 2 O 杂化物显示出从 NO 3 -合成NH 3 的高选择性(96.70%)和法拉第效率(94.32%)。详细表征表明 (1) Pd 能够实现电子转移 (Pd 3d → Cu 3d) 并通过形成部分 PdCu 合金导致 Cu 3d 轨道极化,这使得 Pd 电子缺乏但提供空轨道来吸附 NO 3 -,(2)富电子Cu更有利于NO 3 -还原的发生。在线微分电化学质谱和密度泛函理论计算的相互证实表明,PdCu合金阻止了*NOH中间体的生成并促进了*N的形成,为从NO 3 -还原反应合成NH 3提供了新的机制。










































 京公网安备 11010802027423号
京公网安备 11010802027423号