当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Synthesis of 1-Amino-2,2,2-trifluoroalkylphosphonates from Alkene-Tethered Trifluoroacetimidoyl Chlorides
Organic Letters ( IF 4.9 ) Pub Date : 2021-09-20 , DOI: 10.1021/acs.orglett.1c02750 José F Rodríguez 1 , Anji Zhang 1 , Ramon Arora 1 , Mark Lautens 1
Organic Letters ( IF 4.9 ) Pub Date : 2021-09-20 , DOI: 10.1021/acs.orglett.1c02750 José F Rodríguez 1 , Anji Zhang 1 , Ramon Arora 1 , Mark Lautens 1
Affiliation
|
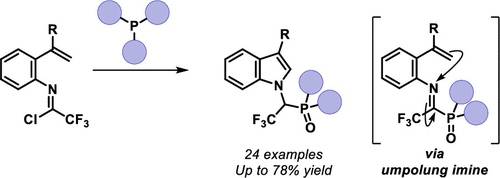
The reaction of alkene-tethered trifluoroacetimidoyl chlorides with trialkyl phosphites furnishes 1-amino-2,2,2-trifluoroalkylphosphonates. The products were generated in moderate to good yields, and the scalability of this process was showcased. Partial hydrolysis of the phosphonate moiety was achieved. The cyclization is proposed to occur via formation of an imidoyl phosphonate intermediate that becomes susceptible to nucleophilic attack at nitrogen through the strong electron-withdrawing groups at the imidoyl carbon.
中文翻译:

从烯烃系三氟乙酰亚胺酰氯合成 1-氨基-2,2,2-三氟烷基膦酸酯
烯烃束缚的三氟乙亚胺酰氯与亚磷酸三烷基酯的反应提供了 1-氨基-2,2,2-三氟烷基膦酸酯。产品以中等到良好的产量生产,并展示了该过程的可扩展性。实现了膦酸酯部分的部分水解。环化反应是通过形成亚氨基膦酸酯中间体而发生的,该中间体通过亚氨酰碳上的强吸电子基团对氮的亲核攻击变得敏感。
更新日期:2021-10-01
中文翻译:

从烯烃系三氟乙酰亚胺酰氯合成 1-氨基-2,2,2-三氟烷基膦酸酯
烯烃束缚的三氟乙亚胺酰氯与亚磷酸三烷基酯的反应提供了 1-氨基-2,2,2-三氟烷基膦酸酯。产品以中等到良好的产量生产,并展示了该过程的可扩展性。实现了膦酸酯部分的部分水解。环化反应是通过形成亚氨基膦酸酯中间体而发生的,该中间体通过亚氨酰碳上的强吸电子基团对氮的亲核攻击变得敏感。











































 京公网安备 11010802027423号
京公网安备 11010802027423号