当前位置:
X-MOL 学术
›
Eur. J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Enantioselective Addition of Sodium Bisulfite to Nitroalkenes. A Convenient Approach to Chiral Sulfonic Acids
European Journal of Organic Chemistry ( IF 2.8 ) Pub Date : 2021-09-15 , DOI: 10.1002/ejoc.202101064 Ehsan Sheikhi 1 , Narjes Rezaei 1 , Alvaro Castilla 1 , Amparo Sanz-Marco 1 , Carlos Vila 1 , M. Carmen Muñoz 2 , Jose R Pedro 1 , Gonzalo Blay 3
European Journal of Organic Chemistry ( IF 2.8 ) Pub Date : 2021-09-15 , DOI: 10.1002/ejoc.202101064 Ehsan Sheikhi 1 , Narjes Rezaei 1 , Alvaro Castilla 1 , Amparo Sanz-Marco 1 , Carlos Vila 1 , M. Carmen Muñoz 2 , Jose R Pedro 1 , Gonzalo Blay 3
Affiliation
|
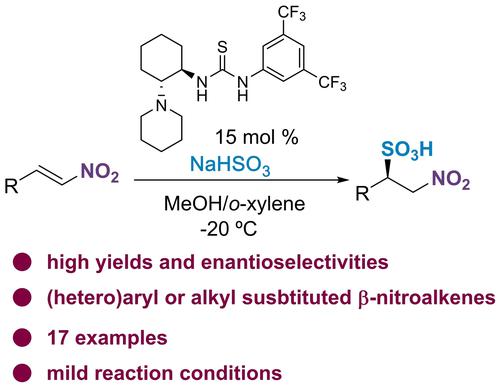
A bifunctional thiourea catalyzes the enantioselective addition of sodium bisulfite to nitroalkenes to give chiral β-nitrosulfonic acids with good yields and enantiomeric excesses. The sulfonic acids can be converted into the corresponding esters by treatment with trimethyl orthoacetate.

中文翻译:

亚硫酸氢钠与硝基烯烃的对映选择性加成。手性磺酸的便捷方法
双功能硫脲催化亚硫酸氢钠与硝基烯烃的对映选择性加成,得到具有良好收率和对映体过量的手性 β-硝基磺酸。通过用原乙酸三甲酯处理可以将磺酸转化为相应的酯。

更新日期:2021-10-06

中文翻译:

亚硫酸氢钠与硝基烯烃的对映选择性加成。手性磺酸的便捷方法
双功能硫脲催化亚硫酸氢钠与硝基烯烃的对映选择性加成,得到具有良好收率和对映体过量的手性 β-硝基磺酸。通过用原乙酸三甲酯处理可以将磺酸转化为相应的酯。




























 京公网安备 11010802027423号
京公网安备 11010802027423号