当前位置:
X-MOL 学术
›
Asian J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Vicinal Trifluoromethylthioamination of Alkenes with Trifluoromethanesulfenamides
Asian Journal of Organic Chemistry ( IF 2.8 ) Pub Date : 2021-08-26 , DOI: 10.1002/ajoc.202100553 Arunachalam Kesavan 1 , Pazhamalai Anbarasan 2
Asian Journal of Organic Chemistry ( IF 2.8 ) Pub Date : 2021-08-26 , DOI: 10.1002/ajoc.202100553 Arunachalam Kesavan 1 , Pazhamalai Anbarasan 2
Affiliation
|
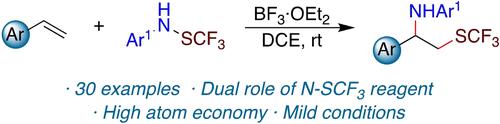
BF3 ⋅ OEt2 meidated vicinal trifluoromethylthioamination of alkenes has been accomplished using trifluoromethanesulfenamides at room temperature. Trifluoromethanesulfenamides play dual role as ‘SCF3’ and amine source, which allows high atom-efficient approach to access 1-trifluoromethylthio-2-aminoalkane derivatives.

中文翻译:

烯烃与三氟甲亚磺酰胺的邻位三氟甲硫胺化
BF 3 ⋅ OEt 2介导的邻位三氟甲硫胺化烯烃已在室温下使用三氟甲磺酰胺完成。三氟甲磺酰胺作为“SCF 3 ”和胺源发挥双重作用,这使得使用高原子效率的方法获得 1-三氟甲硫基-2-氨基烷烃衍生物。

更新日期:2021-10-19

中文翻译:

烯烃与三氟甲亚磺酰胺的邻位三氟甲硫胺化
BF 3 ⋅ OEt 2介导的邻位三氟甲硫胺化烯烃已在室温下使用三氟甲磺酰胺完成。三氟甲磺酰胺作为“SCF 3 ”和胺源发挥双重作用,这使得使用高原子效率的方法获得 1-三氟甲硫基-2-氨基烷烃衍生物。












































 京公网安备 11010802027423号
京公网安备 11010802027423号