当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Enantioselective Lactonization by π-Acid-Catalyzed Allylic Substitution: A Complement to π-Allylmetal Chemistry
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2021-08-22 , DOI: 10.1002/anie.202108336 Arun Raj Kizhakkayil Mangadan 1 , Ji Liu 1 , Aaron Aponick 1
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2021-08-22 , DOI: 10.1002/anie.202108336 Arun Raj Kizhakkayil Mangadan 1 , Ji Liu 1 , Aaron Aponick 1
Affiliation
|
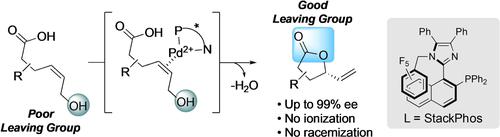
Asymmetric allylic alkylation (AAA) is a powerful method for the formation of highly useful, non-racemic allylic compounds. Here we present a complementary enantioselective process that generates allylic lactones via π-acid catalysis. More specifically, a catalytic enantioselective dehydrative lactonization of allylic alcohols using a novel PdII-catalyst containing the imidazole-based P,N-ligand (S)-StackPhos is reported. The high-yielding reactions are operationally simple to perform with enantioselectivities up to 99 % ee. This strategy facilitates the replacement of a poor leaving group with what would ostensibly be a better leaving group in the product avoiding complications arising from racemization by equilibration.
中文翻译:

π-酸催化烯丙基取代的对映选择性内酯化:π-烯丙基金属化学的补充
不对称烯丙基烷基化 (AAA) 是形成非常有用的非外消旋烯丙基化合物的有效方法。在这里,我们提出了一种互补的对映选择性过程,该过程通过 π 酸催化生成烯丙内酯。更具体地说,报道了使用含有咪唑基P , N-配体 ( S )-StackPhos的新型 Pd II催化剂对烯丙醇进行催化对映选择性脱水内酯化。高产率反应操作简单,对映选择性高达 99 % ee. 该策略有助于用表面上更好的离去基团替换产品中的不良离去基团,避免因平衡外消旋化而引起的并发症。
更新日期:2021-09-27
中文翻译:

π-酸催化烯丙基取代的对映选择性内酯化:π-烯丙基金属化学的补充
不对称烯丙基烷基化 (AAA) 是形成非常有用的非外消旋烯丙基化合物的有效方法。在这里,我们提出了一种互补的对映选择性过程,该过程通过 π 酸催化生成烯丙内酯。更具体地说,报道了使用含有咪唑基P , N-配体 ( S )-StackPhos的新型 Pd II催化剂对烯丙醇进行催化对映选择性脱水内酯化。高产率反应操作简单,对映选择性高达 99 % ee. 该策略有助于用表面上更好的离去基团替换产品中的不良离去基团,避免因平衡外消旋化而引起的并发症。









































 京公网安备 11010802027423号
京公网安备 11010802027423号