当前位置:
X-MOL 学术
›
Eur. J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Versatile Synthesis of Diaminoxylosides via Iodosulfonamidation of Xylal Derivatives
European Journal of Organic Chemistry ( IF 2.5 ) Pub Date : 2021-08-13 , DOI: 10.1002/ejoc.202100792 Sven Ole Jaeschke 1 , Thisbe K. Lindhorst 2
European Journal of Organic Chemistry ( IF 2.5 ) Pub Date : 2021-08-13 , DOI: 10.1002/ejoc.202100792 Sven Ole Jaeschke 1 , Thisbe K. Lindhorst 2
Affiliation
|
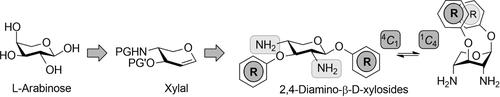
The power of iodosulfonamidation of glycals was used for the synthesis of 2,4-diaminoxylosides, which are of interest as conformational switches. The synthetic pathway proceeds from an l-arabino configuration over a glycal with two stereocenters in a threo relation to the d-xylo configuration of the product with compelling selectivity.

中文翻译:

通过木醛衍生物的碘磺酰胺化多功能合成二氨基木糖苷
糖基的碘磺酰胺化作用用于合成 2,4-二氨基木糖苷,其作为构象开关很受关注。从该合成途径进行升-阿拉伯糖在具有在两个立构中心的构型的烯糖苏相对于d -木糖与引人注目选择性的产品的构造。

更新日期:2021-08-13

中文翻译:

通过木醛衍生物的碘磺酰胺化多功能合成二氨基木糖苷
糖基的碘磺酰胺化作用用于合成 2,4-二氨基木糖苷,其作为构象开关很受关注。从该合成途径进行升-阿拉伯糖在具有在两个立构中心的构型的烯糖苏相对于d -木糖与引人注目选择性的产品的构造。












































 京公网安备 11010802027423号
京公网安备 11010802027423号