Journal of Industrial and Engineering Chemistry ( IF 5.9 ) Pub Date : 2021-07-16 , DOI: 10.1016/j.jiec.2021.07.013 Liying Song 1 , Hao Shi 1 , Peng Han 2 , Xiaohong Ji 3 , Fubin Ma 3
|
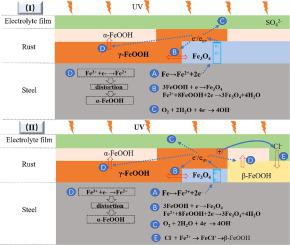
A contrastive study about ultraviolet light (UV) illumination on Na2SO4, NaCl induced Q450 weathering steel was qualitatively and quantitatively studied. The corrosion rate was calculated from mass losses and the order was as following, coupons deposited with Na2SO4 under UV illumination (238.18 μA·cm−2) > with NaCl under UV illumination (213.77 μA·cm−2) > with NaCl under Dark condition (71.98 μA·cm−2) > with Na2SO4 under Dark condition (43.26 μA·cm−2). The influence of UV illumination on metal corrosion followed this sequence: Na2SO4 > NaCl, and which was on account of corrosion products film on metal depositing Na2SO4 with a larger carrier density on metal depositing Na2SO4 with a larger carrier density (9.321020 cm−3) and stronger electric conductivity ability than NaCl (8.931020 cm−3). Electrochemical impedance spectroscopy studies revealed charge transfer resistance Rct value, suggesting that smaller resistances gained on the rusted samples due to UV illumination, and an accelerated corrosion effect on rusted sample was also obtained under UV illumination. The improved corrosion performance by UV illumination was due to corrosion products of semiconductor property and photovoltaic effect.
中文翻译:

紫外光照射下Na2SO4、NaCl沉积的Q450耐候钢的腐蚀趋势
对Na 2 SO 4、NaCl诱导的Q450耐候钢的紫外光(UV)照射进行了定性和定量对比研究。腐蚀速率由质量损失计算,顺序如下,在紫外光照射下用 Na 2 SO 4沉积(238.18 μA·cm -2)>在紫外光照射下用氯化钠(213.77 μA·cm -2)>用氯化钠在黑暗条件下 (71.98 μA·cm -2 ) >在黑暗条件下使用 Na 2 SO 4 (43.26 μA·cm -2 )。紫外线照射对金属腐蚀的影响遵循以下顺序: Na 2 SO 4 > NaCl,这是由于在金属上沉积具有较大载流子密度的Na 2 SO 4的腐蚀产物膜,在金属上沉积具有较大载流子密度的 Na 2 SO 4 (9.3210 20 cm -3 ) 和比 NaCl (8.9310 20厘米-3)。电化学阻抗谱研究揭示了电荷转移电阻R ct值,表明由于紫外线照射在生锈样品上获得的电阻较小,并且在紫外线照射下也获得了对生锈样品的加速腐蚀作用。UV 照射改善的腐蚀性能是由于半导体性质和光伏效应的腐蚀产物。









































 京公网安备 11010802027423号
京公网安备 11010802027423号