当前位置:
X-MOL 学术
›
Eur. J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Spiro‐2‐oxindoles via 1,3‐dipolar cycloadditions. A decade update
European Journal of Organic Chemistry ( IF 2.5 ) Pub Date : 2021-02-23 , DOI: 10.1002/ejoc.202100121 Giorgio Molteni 1 , Alessandra Silvani 2
European Journal of Organic Chemistry ( IF 2.5 ) Pub Date : 2021-02-23 , DOI: 10.1002/ejoc.202100121 Giorgio Molteni 1 , Alessandra Silvani 2
Affiliation
|
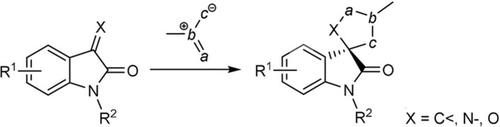
The reactivity of 2‐oxindoles bearing a C=X double bond (X=C<, N−, O) in the 3‐position towards a variety of 1,3‐dipolar species has been reviewed in a systematic way according to the type of the 1,3‐dipole, the 2‐oxindole dipolarophile, and the racemic or asymmetric nature of the cycloaddition. Because of the great variety of the resulting spiro‐2‐oxindole skeletons, the developments occurred in the 2010–2020 decade were taken into account.

中文翻译:

通过1,3-偶极环加成得到的螺-2-氧吲哚。十年更新
根据类型,系统地审查了在3位带有C = X双键(X = C <,N-,O)的2-氧吲哚对各种1,3-偶极物种的反应性1,3-偶极,2-氧吲哚偶极亲和性和环加成的外消旋或不对称性质。由于产生的螺2-2-氧吲哚骨架种类繁多,因此将2010–2020十年间的发展情况考虑在内。

更新日期:2021-03-15

中文翻译:

通过1,3-偶极环加成得到的螺-2-氧吲哚。十年更新
根据类型,系统地审查了在3位带有C = X双键(X = C <,N-,O)的2-氧吲哚对各种1,3-偶极物种的反应性1,3-偶极,2-氧吲哚偶极亲和性和环加成的外消旋或不对称性质。由于产生的螺2-2-氧吲哚骨架种类繁多,因此将2010–2020十年间的发展情况考虑在内。












































 京公网安备 11010802027423号
京公网安备 11010802027423号