Medicinal Chemistry ( IF 1.9 ) Pub Date : 2021-02-28 , DOI: 10.2174/1573406416666200610191002 Uma Krithika 1 , Prabitha P 1 , Subhankar P. Mandal 1 , Yuvaraj S 1 , Priya D 1 , Ashish D Wadhwani 2 , B. R. Prashantha Kumar 1
|
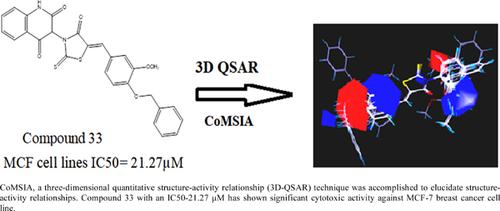
Background: A series of novel 5-substituted benzylidene rhodanine derivatives using four different amines were designed based on our previously developed CoMSIA (Comparative molecular similarity indices analysis) model for the anticancer activity.
Methods: The designed rhodanines were synthesized via dithiocarbamate formation, cyclization and Knoevenagel condensation. The structures of the synthesized compounds were confirmed and analyzed by spectral studies.
Results: The synthesized rhodanines were investigated for in vitro anticancer activities and the analogs have displayed mild to significant cytotoxicity against MCF-7 breast cancer cells. The compounds with benzyloxy substitution at the fifth position of rhodanine ring (Compounds 20, 33 and 38) system showed significant cytotoxic activity against MCF-7 cells. CoMSIA, a three-dimensional quantitative structureactivity relationship (3D-QSAR) technique was accomplished to elucidate structure-activity relationships.
Conclusion: Based on the information derived from CoMSIA contour plots, some key features for increasing the activity of compounds have been identified and used to design new anti-cancer agents. The present developed CoMSIA model displayed good external predictability, r2pred of 0.841 and good statistical robustness.
中文翻译:

新型罗丹宁类似物作为抗癌药的开发:设计,合成,评估和CoMSIA研究
背景:基于我们以前开发的CoMSIA(比较分子相似性指数分析)模型,设计了使用四种不同胺的一系列新型5-取代亚苄基罗丹宁衍生物。
方法:通过二硫代氨基甲酸酯的形成,环化和Knoevenagel缩合合成所设计的罗丹酮。通过光谱研究证实并分析了合成化合物的结构。
结果:研究了合成的罗丹丹的体外抗癌活性,其类似物对MCF-7乳腺癌细胞显示出轻度至显着的细胞毒性。在若丹宁环的第五位(化合物20、33和38)系统具有苄氧基取代的化合物显示出对MCF-7细胞的明显细胞毒活性。CoMSIA是一种三维定量结构活性关系(3D-QSAR)技术,用于阐明结构活性关系。
结论:根据从CoMSIA等高线图获得的信息,已确定了增加化合物活性的一些关键特征,并将其用于设计新的抗癌药。目前开发的CoMSIA模型显示出良好的外部可预测性,r2pred为0.841和良好的统计稳健性。











































 京公网安备 11010802027423号
京公网安备 11010802027423号