Separation and Purification Technology ( IF 8.1 ) Pub Date : 2021-01-15 , DOI: 10.1016/j.seppur.2021.118335 Hongbin Wang , Jiewen Deng , Xiaohui Lu , Li Wan , Jin Huang , Yiqing Liu
|
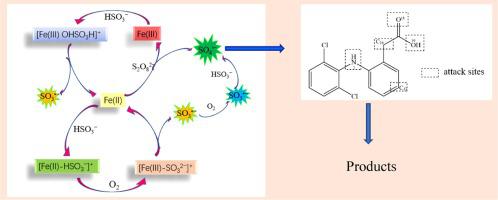
The improvement on diclofenac (DCF) degradation by Fe(II)-activated persulfate (PS) combined with bisulfite (BS) was investigated. It was observed that the introduction of BS in Fe(II)/PS process enhanced the circulation of Fe(III/II) and kept Fe(II) at a high concentration, resulting a rapid and continuous DCF degradation. The initial pH, reagent dosages and reaction temperature played important roles on DCF removal in BS/Fe(II)/PS process. The main iron species in this system were FeHSO3+, Fe2+ and FeOHSO3H+, and their variation trends were calculated by the software of MINITEQ 3.1. Based on the alcohols quenching experiments, SO4 − was confirmed to be the prominent reactive radical for DCF oxidation in BS/Fe(II)/PS process. Moreover, the probable DCF degradation mechanism was proposed based on the detected intermediate products and density functional theory (DFT) calculation. The BS/Fe(II)/PS system presented a synergistic effect for rapid and steady DCF degradation through the occurred cycle of Fe(II/III) (i.e., oxidation of Fe(II) by PS and then reduction of Fe(III) by BS) and solved the drawbacks of slow initial reaction of Fe(II)/BS and weak durability of Fe(II)/PS. This study could provide an effective and environmentally friendly approach to sewage treatment.
− was confirmed to be the prominent reactive radical for DCF oxidation in BS/Fe(II)/PS process. Moreover, the probable DCF degradation mechanism was proposed based on the detected intermediate products and density functional theory (DFT) calculation. The BS/Fe(II)/PS system presented a synergistic effect for rapid and steady DCF degradation through the occurred cycle of Fe(II/III) (i.e., oxidation of Fe(II) by PS and then reduction of Fe(III) by BS) and solved the drawbacks of slow initial reaction of Fe(II)/BS and weak durability of Fe(II)/PS. This study could provide an effective and environmentally friendly approach to sewage treatment.
中文翻译:

Fe(II)活化的过硫酸盐和亚硫酸氢盐快速连续降解双氯芬酸
研究了Fe(II)活化的过硫酸盐(PS)结合亚硫酸氢盐(BS)对双氯芬酸(DCF)降解的改善。观察到,在Fe(II)/ PS工艺中引入BS增强了Fe(III / II)的循环,并使Fe(II)保持在高浓度,从而导致DCF快速连续地降解。初始pH,试剂用量和反应温度对BS / Fe(II)/ PS工艺中DCF的去除起重要作用。该系统中的主要铁种类为FeHSO 3 +,Fe 2+和FeOHSO 3 H +,并通过MINITEQ 3.1软件计算了它们的变化趋势。基于该醇猝灭实验,SO 4  -被证实是BS / Fe(II)/ PS工艺中DCF氧化的主要反应自由基。此外,基于检测到的中间产物和密度泛函理论(DFT)计算,提出了可能的DCF降解机理。BS / Fe(II)/ PS系统通过发生的Fe(II / III)循环(即PS氧化Fe(II)然后还原Fe(III))呈现了快速稳定DCF降解的协同效应。解决了Fe(II)/ BS的初始反应缓慢和Fe(II)/ PS的耐久性差的缺点。这项研究可以为污水处理提供一种有效且环保的方法。
-被证实是BS / Fe(II)/ PS工艺中DCF氧化的主要反应自由基。此外,基于检测到的中间产物和密度泛函理论(DFT)计算,提出了可能的DCF降解机理。BS / Fe(II)/ PS系统通过发生的Fe(II / III)循环(即PS氧化Fe(II)然后还原Fe(III))呈现了快速稳定DCF降解的协同效应。解决了Fe(II)/ BS的初始反应缓慢和Fe(II)/ PS的耐久性差的缺点。这项研究可以为污水处理提供一种有效且环保的方法。











































 京公网安备 11010802027423号
京公网安备 11010802027423号