当前位置:
X-MOL 学术
›
Org. Chem. Front.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Photochemical synthesis of 3-hydroxyphenanthro[9,10-c]furan-1(3H)-ones from α-keto acids and alkynes
Organic Chemistry Frontiers ( IF 4.6 ) Pub Date : 2021-1-4 , DOI: 10.1039/d0qo01487g Beibei Zhao 1, 2, 3, 4, 5 , Zhen Zhang 1, 2, 3, 4, 5 , Yu Ge 1, 2, 3, 4, 5 , Pinhua Li 1, 2, 3, 4, 5 , Tao Miao 1, 2, 3, 4, 5 , Lei Wang 1, 2, 3, 4, 5
Organic Chemistry Frontiers ( IF 4.6 ) Pub Date : 2021-1-4 , DOI: 10.1039/d0qo01487g Beibei Zhao 1, 2, 3, 4, 5 , Zhen Zhang 1, 2, 3, 4, 5 , Yu Ge 1, 2, 3, 4, 5 , Pinhua Li 1, 2, 3, 4, 5 , Tao Miao 1, 2, 3, 4, 5 , Lei Wang 1, 2, 3, 4, 5
Affiliation
|
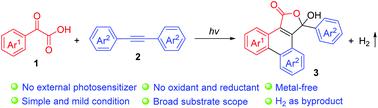
A novel and efficient method for the synthesis of 3-hydroxyphenanthro[9,10-c]furan-1(3H)-ones has been achieved from readily available α-keto acids and alkynes through photo-initiated transformation, providing a range of structurally diverse products in good to excellent yields under mild conditions. Mechanistic studies reveal that this reaction undergoes photo-induced radical addition/cyclization and hydrogen evolution cascade processes.
中文翻译:

由α-酮酸和炔烃光化学合成3-羟基菲咯啉[9,10-c]呋喃-1(3H)-一
通过光引发的转化,从容易获得的α-酮酸和炔烃中获得了一种新颖且有效的合成3-羟基菲[9,10- c ]呋喃-1(3 H)-的方法,提供了一系列在温和的条件下,结构多样的产品,收率好至极好。机理研究表明,该反应经历了光诱导的自由基加成/环化和氢释放级联过程。
更新日期:2021-01-14
中文翻译:

由α-酮酸和炔烃光化学合成3-羟基菲咯啉[9,10-c]呋喃-1(3H)-一
通过光引发的转化,从容易获得的α-酮酸和炔烃中获得了一种新颖且有效的合成3-羟基菲[9,10- c ]呋喃-1(3 H)-的方法,提供了一系列在温和的条件下,结构多样的产品,收率好至极好。机理研究表明,该反应经历了光诱导的自由基加成/环化和氢释放级联过程。











































 京公网安备 11010802027423号
京公网安备 11010802027423号