Journal of Molecular Liquids ( IF 5.3 ) Pub Date : 2021-01-12 , DOI: 10.1016/j.molliq.2021.115366 Ivaylo Tankov , Rumyana Yankova
|
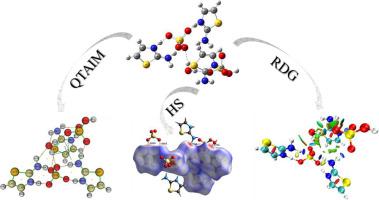
Nature of the chemical bonds and intermolecular interactions in the multifunctional ionic liquid tris(2-aminothiazolium) hydrogen sulfate sulfate monohydrate ([tr(2-AT]+[HSO4]−.SO42−.H2O) are investigated in the current work by means of a density functional theory at B3LYP/6–311++G(d,p) level. For that purpose, detailed structural, natural bond orbital (NBO), Hirshfeld surface (HS) and reduced density gradient (RDG) analyses are employed. In addition, the Quantum theory of atoms in molecules (QTAIM) is applied as well. Structural, NBO and HS analyses showed that the bond lengths in the title compound depend strongly on the intermolecular O···H interactions between amine ((N H)am) and thiazole ((N
H)am) and thiazole ((N H)thzl) hydrogen atoms in cationic ([tr(2-AT]+) fragments with oxygen atoms in anionic ([HSO4]− and SO42−) and H2O moieties. Moreover, hydrogen bonds between H2O with [HSO4]− and SO42− are also observed. QTAIM and RDG data revealed that the (N
H)thzl) hydrogen atoms in cationic ([tr(2-AT]+) fragments with oxygen atoms in anionic ([HSO4]− and SO42−) and H2O moieties. Moreover, hydrogen bonds between H2O with [HSO4]− and SO42− are also observed. QTAIM and RDG data revealed that the (N H)am···(O
H)am···(O S)s and (N
S)s and (N H)thzl···(O
H)thzl···(O S)s bonds are partly covalent in nature. In contrary, the interactions (N
S)s bonds are partly covalent in nature. In contrary, the interactions (N H)am···(O
H)am···(O S)hs, (N
S)hs, (N H)thzl···(O
H)thzl···(O S)hs and (N
S)hs and (N H)am···(O
H)am···(O H)w are define as electrostatic, being observed in a greater extent for (N
H)w are define as electrostatic, being observed in a greater extent for (N H)am···(O
H)am···(O H)w.
H)w.
中文翻译:

多功能离子液体三(2-氨基噻唑)硫酸氢硫酸盐一水合物中化学相互作用的性质
研究了多功能离子液体三(2-氨基噻唑)硫酸氢一水合硫酸盐([tr(2-AT] + [HSO 4 ] -. SO 4 2− .H 2 O)中化学键的性质和分子间的相互作用。目前的工作是通过B3LYP / 6–311 ++ G(d,p)级的密度泛函理论进行的。为此,详细的结构,自然键轨道(NBO),Hirshfeld表面(HS)和降低的密度梯度(使用RDG)分析,此外,还应用了分子中的量子理论(QTAIM),结构,NBO和HS分析表明标题化合物中的键长强烈依赖于分子间的O···H相互作用胺之间(( NH)am)和噻唑(([N
NH)am)和噻唑(([N  H] thzl)氢原子在阳离子([tr(2-AT] +)片段中带有氧原子,在阴离子([HSO 4 ] -和SO 4 2-]中)和H 2 O部分。 ,H之间的氢键2 ö与[HSO 4 ] -和SO 4 2-。还观察到QTAIM和RDG数据显示,该(N
H] thzl)氢原子在阳离子([tr(2-AT] +)片段中带有氧原子,在阴离子([HSO 4 ] -和SO 4 2-]中)和H 2 O部分。 ,H之间的氢键2 ö与[HSO 4 ] -和SO 4 2-。还观察到QTAIM和RDG数据显示,该(N  1H)是...(O
1H)是...(O  S)小号和(N
S)小号和(N  1H)thzl ·· ·(OS
1H)thzl ·· ·(OS  )s键本质上是部分共价的。相反,相互作用(N
)s键本质上是部分共价的。相反,相互作用(N H)am ···(O
H)am ···(O  S)hs,(NH
S)hs,(NH )thzl···(O
)thzl···(O  S)hs和(NH
S)hs和(NH )am ···(O
)am ···(O  H)w定义为静电,在更大程度上被观察到对于(N
H)w定义为静电,在更大程度上被观察到对于(N  H)am ···(OH
H)am ···(OH  )w。
)w。











































 京公网安备 11010802027423号
京公网安备 11010802027423号