Current Organic Chemistry ( IF 1.7 ) Pub Date : 2020-11-30 , DOI: 10.2174/1385272824999201020204001 Dukanya 1 , Toreshettahally R. Swaroop 1 , Kanchugarakoppal S. Rangappa 2 , Basappa 1
|
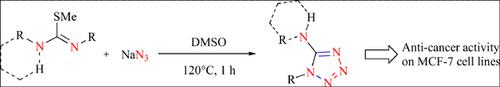
Synthesis of 1,5-disubstituted tetrazoles by the cyclization of sodium azide with N(N),N'-di(tri)substituted carbamimidothioate is reported. Tetrazoles are obtained in good to excellent yield in the absence of a catalyst. All the compounds were characterized by NMR and HRMS analysis. Single crystal X-ray diffraction data of 1-(4-chlorophenyl)-4-(5-phenyl- 1H-tetrazol-1-yl)piperazine 5g is also provided. Further, these disubstituted tetrazoles were tested against the proliferation of human breast cancer cells (MCF-7), which identified 5e as a lead compound. Finally, we have shown in silico that these compounds may interact with the ligand binding domain of estrogen receptor α (ERα), that expresses at high amount in MCF-7 cells.
中文翻译:

N(N),N'-二(三)取代的氨基甲硫基甲基甲烷叠氮化钠的环缩合反应:1,5-二取代的四唑的合成及其对人乳腺癌细胞的细胞毒性的新维度
据报道,通过叠氮化钠与N(N),N′-二(三)取代的氨基甲酰胺基硫代酸酯的环化反应合成1,5-二取代的四唑。在不存在催化剂的情况下,以良好或优异的产率获得四唑。通过NMR和HRMS分析对所有化合物进行表征。还提供了1-(4-氯苯基)-4-(5-苯基-1H-四唑-1-基)哌嗪5g的单晶X射线衍射数据。此外,测试了这些二取代的四唑类抗人类乳腺癌细胞(MCF-7)的增殖,后者确定5e为先导化合物。最后,我们在计算机上显示了这些化合物可能与雌激素受体α(ERα)的配体结合域相互作用,后者在MCF-7细胞中大量表达。











































 京公网安备 11010802027423号
京公网安备 11010802027423号