Tetrahedron Letters ( IF 1.8 ) Pub Date : 2020-12-05 , DOI: 10.1016/j.tetlet.2020.152689 Balu Misal , Amey Palav , Prerna Ganwir , Ganesh Chaturbhuj
|
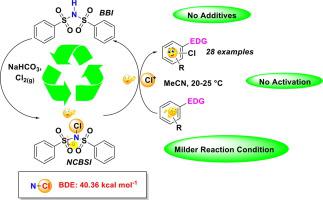
N-Chloro-N-(phenylsulfonyl)benzene sulfonamide (NCBSI) has been explored for the first time as a chlorinating reagent for direct chlorination of various activated arenes and heterocycles without any activator. A comparative in-silico study was performed to determine the electrophilic character for NCBSI and commercially available N-chloro reagents to reveal the reactivity on a theoretical viewpoint. The reagent was prepared by an improved method avoiding the use of hazardous t-butyl hypochlorite. This reagent was proved to be very reactive compared to other N-chloro reagents. The precursor of the reagent N-(phenylsulfonyl)benzene sulfonamide was recovered from aqueous spent, which can be recycled to synthesize NCBSI. The eco-friendly protocol was equally applicable for the synthesis of industrially important chloroxylenol as an antibacterial agent.
中文翻译:

N-氯-N-(苯磺酰基)苯磺酰胺(NCBSI)对活化的芳烃进行无活化剂的快速,环保的氯化反应
N-氯-N-(苯磺酰基)苯磺酰胺(NCBSI)首次被开发为无需任何活化剂即可直接氯化各种活化的芳烃和杂环的氯化试剂。的比较中,计算机芯片上进行研究,以确定NCBSI亲电性质和市售Ñ氯试剂,以揭示理论的角度来看的反应性。通过避免使用有害的次氯酸叔丁基酯的改进方法制备了该试剂。与其他N-氯试剂相比,该试剂具有很高的反应活性。试剂N的前体从废废水中回收-(苯磺酰基)苯磺酰胺,可将其再循环以合成NCBSI。环保协议同样适用于工业上重要的氯氧亚麻酚作为抗菌剂的合成。



























 京公网安备 11010802027423号
京公网安备 11010802027423号