European Journal of Medicinal Chemistry ( IF 6.0 ) Pub Date : 2020-12-05 , DOI: 10.1016/j.ejmech.2020.113087 Michael Moir , Samuel Lane , Andrew P. Montgomery , David Hibbs , Mark Connor , Michael Kassiou
|
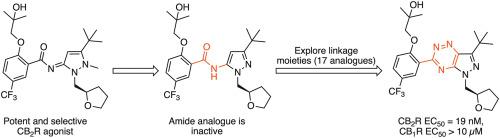
The development of selective CB2 receptor agonists is a promising therapeutic approach for the treatment of inflammatory diseases, without CB1 receptor mediated psychoactive side effects. Preliminary structure-activity relationship studies on pyrazoylidene benzamide agonists revealed the -ylidene benzamide moiety was crucial for functional activity at the CB2 receptor. A small library of compounds with varying linkage moieties between the pyrazole and substituted phenyl group has culminated in the discovery of a potent and selective pyrazolo-[2,3-e]-[1,2,4]-triazine agonist 19 (CB2R EC50 = 19 nM, CB1R EC50 > 10 μM). Docking studies have revealed key structural features of the linkage group that are important for potent functional activity.
中文翻译:

有效和选择性的吡唑并-[2,3- e ]-[1,2,4]-三嗪大麻素2型受体激动剂的发现
选择性CB 2受体激动剂的开发是一种有希望的治疗炎性疾病的治疗方法,没有CB 1受体介导的精神活性副作用。对吡唑基苯甲酰胺激动剂的初步结构-活性关系研究表明,亚苄基苯甲酰胺部分对于CB 2受体的功能活性至关重要。在吡唑和取代的苯基之间具有不同连接部分的小型化合物文库最终发现了有效的选择性吡唑并-[2,3- e ]-[1,2,4]-三嗪激动剂19(CB 2 R EC 50 = 19 nM,CB 1 R EC 50 > 10μM)。对接研究已经揭示了该连接基团的关键结构特征,这些特征对于有效的功能活性很重要。











































 京公网安备 11010802027423号
京公网安备 11010802027423号