Bioorganic & Medicinal Chemistry ( IF 3.5 ) Pub Date : 2020-11-18 , DOI: 10.1016/j.bmc.2020.115877 Ryan P Downs 1 , Zhousheng Xiao 2 , Munachi O Ikedionwu 1 , Jacob W Cleveland 1 , Ai Lin Chin 1 , Abigail E Cafferty 1 , L Darryl Quarles 2 , Jesse D Carrick 1
|
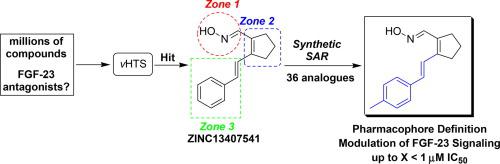
Hereditary hypophosphatemic disorders, TIO, and CKD conditions are believed to be influenced by an excess of Fibroblast Growth Factor-23 (FGF-23) which activates a binary renal FGFRs / α-Klotho complex to regulate homeostatic metabolism of phosphate and vitamin D. Adaptive FGF-23 responses from CKD patients with excess FGF-23 frequently lead to increased mortality from cardiovascular disease. A reversibly binding small molecule therapeutic has yet to emerge from research and development in this area. Current outcomes described in this work highlight efforts related to lead identification and modification using organic synthesis of strategic analogues to probe structure-activity relationships and preliminarily define the pharmacophore of a computationally derived hit obtained from virtual high-throughput screening. Synthetic strategies for the initial hit and analogue preparation, as well as preliminary cellular in vitro assay results highlighting sub micromolar inhibition of the FGF-23 signaling sequence at a concentration well below cytotoxicity are reported herein.
中文翻译:

FGF-23 拮抗剂的设计和开发:合成类似物探测的药效团和初始构效关系的定义
遗传性低磷血症、TIO 和 CKD 病症被认为受过量成纤维细胞生长因子 23 (FGF-23) 的影响,该因子激活二元肾 FGFR/α-Klotho 复合物以调节磷酸盐和维生素 D 的稳态代谢。 适应性来自具有过量 FGF-23 的 CKD 患者的 FGF-23 反应经常导致心血管疾病死亡率增加。在该领域的研究和开发中尚未出现可逆结合的小分子疗法。这项工作中描述的当前结果突出了与使用战略类似物的有机合成来探测结构-活性关系并初步定义从虚拟高通量筛选中获得的计算衍生命中的药效团的先导识别和修饰相关的工作。本文报道了在远低于细胞毒性的浓度下对 FGF-23 信号序列的亚微摩尔抑制的体外测定结果。



























 京公网安备 11010802027423号
京公网安备 11010802027423号