当前位置:
X-MOL 学术
›
Catal. Sci. Technol.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Zirconium and hafnium polyhedral oligosilsesquioxane complexes – green homogeneous catalysts in the formation of bio-derived ethers via a MPV/etherification reaction cascade
Catalysis Science & Technology ( IF 4.4 ) Pub Date : 2020-10-23 , DOI: 10.1039/d0cy01864c Shipra Garg 1, 2, 3, 4 , Daniel K. Unruh 1, 2, 3, 4 , Clemens Krempner 1, 2, 3, 4
Catalysis Science & Technology ( IF 4.4 ) Pub Date : 2020-10-23 , DOI: 10.1039/d0cy01864c Shipra Garg 1, 2, 3, 4 , Daniel K. Unruh 1, 2, 3, 4 , Clemens Krempner 1, 2, 3, 4
Affiliation
|
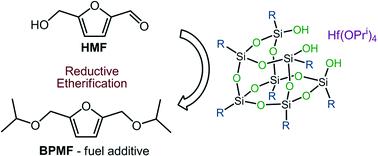
The polyhedral oligosilsesquioxane complexes, {[(isobutyl)7Si7O12]ZrOPri·(HOPri)}2 (I), {[(cyclohexyl)7Si7O12]ZrOPri·(HOPri)}2 (II), {[(isobutyl)7Si7O12]HfOPri·(HOPri)}2 (III) and {[(cyclohexyl)7Si7O12]HfOPri·(HOPri)}2 (IV), were synthesized in good yields from the reactions of M(OPri)4 (M = Zr, Hf) with R-POSS(OH)3 (R = isobutyl, cyclohexyl), resp. I–IV were characterized by 1H, 13C and 29Si NMR spectroscopy and their dimeric solid-state structures were confirmed by X-ray analysis. I–IV catalyze the reductive etherification of 2-hydroxy- and 4-hydroxy and 2-methoxy and 4-methoxybenzaldehyde and vanillin to their respective isopropyl ethers in isopropanol as a “green” solvent and reagent. I–IV are durable and robust homogeneous catalysts operating at temperatures of 100–160 °C for days without significant loss of catalytic activity. Likewise, I–IV selectively catalyze the conversion of 5-hydroxymethylfurfural (HMF) into 2,5-bis(isopropoxymethyl)furane (BPMF), a potentially high-performance fuel additive. Similar results were achieved by using a combination of M(OPri)4 and ligand R-POSS(OH)3 as a catalyst system demonstrating the potential of this “in situ” approach for applications in biomass transformations. A tentative reaction mechanism for the reductive etherification of aldehydes catalysed by I–IV is proposed.
中文翻译:

锆和ha多面体低聚倍半硅氧烷复合物–通过MPV /醚化反应级联形成生物衍生醚的绿色均相催化剂
多面体低聚倍半硅氧烷络合物{[((异丁基)7 Si 7 O 12 ] ZrOPr i ·(HOPr i)}} 2(I),{[(环己基)7 Si 7 O 12 ] ZrOPr i ·(HOPr i)} 2(II),{[((异丁基)7 Si 7 O 12 ] HfOPr i ·(HOPr i)}} 2(III)和{[(环己基)7 Si 7 O 12 ] HfOPr i·(HOPr i)} 2(IV),是由M(OPr i)4(M = Zr,Hf)与R-POSS(OH)3(R =异丁基,环己基)反应合成的,收率很高。I – IV用1 H,13 C和29 Si NMR光谱表征,其二聚体固态结构通过X射线分析证实。I – IV在异丙醇中催化2-羟基和4-羟基,2-甲氧基和4-甲氧基苯甲醛和香兰素还原成醚化成各自的异丙醚,作为“绿色”溶剂和试剂。一世– IV是耐用且坚固的均相催化剂,可在100–160°C的温度下运行几天,而不会显着降低催化活性。同样,I – IV选择性催化5-羟甲基糠醛(HMF)转化为2,5-双(异丙氧基甲基)呋喃(BPMF),这是一种潜在的高性能燃料添加剂。通过使用M(OPr i)4和配体R-POSS(OH)3的组合作为催化剂体系,获得了类似的结果,证明了这种“原位”方法在生物质转化中的应用潜力。I - IV催化醛还原醚化的初步反应机理 被提议。
更新日期:2020-11-17
中文翻译:

锆和ha多面体低聚倍半硅氧烷复合物–通过MPV /醚化反应级联形成生物衍生醚的绿色均相催化剂
多面体低聚倍半硅氧烷络合物{[((异丁基)7 Si 7 O 12 ] ZrOPr i ·(HOPr i)}} 2(I),{[(环己基)7 Si 7 O 12 ] ZrOPr i ·(HOPr i)} 2(II),{[((异丁基)7 Si 7 O 12 ] HfOPr i ·(HOPr i)}} 2(III)和{[(环己基)7 Si 7 O 12 ] HfOPr i·(HOPr i)} 2(IV),是由M(OPr i)4(M = Zr,Hf)与R-POSS(OH)3(R =异丁基,环己基)反应合成的,收率很高。I – IV用1 H,13 C和29 Si NMR光谱表征,其二聚体固态结构通过X射线分析证实。I – IV在异丙醇中催化2-羟基和4-羟基,2-甲氧基和4-甲氧基苯甲醛和香兰素还原成醚化成各自的异丙醚,作为“绿色”溶剂和试剂。一世– IV是耐用且坚固的均相催化剂,可在100–160°C的温度下运行几天,而不会显着降低催化活性。同样,I – IV选择性催化5-羟甲基糠醛(HMF)转化为2,5-双(异丙氧基甲基)呋喃(BPMF),这是一种潜在的高性能燃料添加剂。通过使用M(OPr i)4和配体R-POSS(OH)3的组合作为催化剂体系,获得了类似的结果,证明了这种“原位”方法在生物质转化中的应用潜力。I - IV催化醛还原醚化的初步反应机理 被提议。











































 京公网安备 11010802027423号
京公网安备 11010802027423号