Synthesis ( IF 2.2 ) Pub Date : 2020-11-12 , DOI: 10.1055/s-0040-1705969 Yang Wang , Zheng-Guang Wu , Bing Yang , Xin-Xing Wu
|
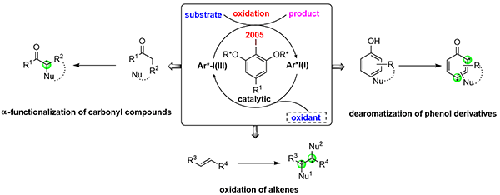
Chiral iodoarenes have been steadily increasing in importance in recent years, especially in enantioselective synthesis and catalysis. Since the development of the concept of chiral iodine(I/III) catalysis, the use of various chiral aryliodine frameworks has been explored in this area. This short review gives an overview of the use of chiral hypervalent iodine(I/III) reagents based on the 2-iodoresorcinol core with two attached two lactic side chains bearing ester or amide groups for the catalytic enantioselective dearomatization of phenol compounds, asymmetric oxidation of alkenes, and enantioselective α-functionalization of carbonyl compounds highlighting the excellent reactivities in terms of yield and enantioselectivity.
1 Introduction
2 Enantioselective Dearomatization of Phenol Derivatives
3 Asymmetric Oxidation of Alkenes
4 Enantioselective α-Functionalization of Carbonyl Compounds
5 Conclusion and Outlook
中文翻译:

基于2-异戊二烯醇核的手性丙烯醛在不对称催化中的最新应用
近年来,手性碘代芳烃的重要性一直在稳步提高,特别是在对映选择性合成和催化方面。自从手性碘(I / III)催化概念的发展以来,在这一领域中已探索了各种手性阿里碘的骨架的使用。这篇简短的综述概述了使用基于2-异戊二烯核心的手性高价碘(I / III)试剂,其中两个带有两个带有酯或酰胺基的乳酸侧链,用于酚类化合物的催化对映选择性脱芳香化,烯烃和羰基化合物的对映选择性α-官能化突出了在收率和对映选择性方面的出色反应性。
1引言
2苯酚衍生物的对映选择性脱芳香化
3烯烃的不对称氧化
4羰基化合物的对映选择性α-官能化
5结论与展望











































 京公网安备 11010802027423号
京公网安备 11010802027423号