Bioorganic & Medicinal Chemistry ( IF 3.3 ) Pub Date : 2020-11-08 , DOI: 10.1016/j.bmc.2020.115848 Jing-Tao Zhang 1 , Li-Xia Wang 2 , Feng-Min Yang 2 , Luo Yang 3 , Yan Liu 2 , Ya-Lin Tang 4
|
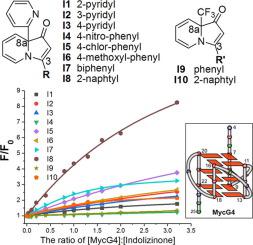
Owing to its potential biological relevance, DNA G-quadruplex has been considered as a prospective anti-cancer target. Some known G-quadruplex-interactive N-containing compounds with low cytotoxicity have become prospective anticancer drugs. Here we reported a new type of N-containing alkaloids 3,8a-disubstituted indolizinones, and investigated their substituent effects at 3- and 8a-positions in targeting to DNA c-myc G-quadruplex. And then we used 3-naphtyl-8a-(pyridin-2-yl) substrate I8 as an example, and investigated its ability in targeting to DNA parallel G-quadruplexes in vitro.
中文翻译:

3,8a-二取代吲哚嗪酮选择性识别DNA平行G-四链体
由于其潜在的生物学相关性,DNA G-四链体已被认为是一种潜在的抗癌靶点。一些已知的G-四链交互式Ñ含具有低细胞毒性的化合物已成为潜在的抗癌药物。在这里,我们报道了一种新型的Ñ含生物碱3,8a二取代indolizinones,并且在靶向DNA c-myc的G-四链体在3-和8a位上研究了它们的取代基的影响。然后我们以3-naphtyl-8a-(pyridin-2-yl)底物I8为例,研究了其体外靶向DNA平行G-四链体的能力。











































 京公网安备 11010802027423号
京公网安备 11010802027423号