Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Photoinduced [3+2] Annulation of Alkene with o-Iodoanilines: An Expedient Approach to Indolines
Synthesis ( IF 2.2 ) Pub Date : 2020-11-04 , DOI: 10.1055/s-0040-1705963 Wujiong Xia , Xinxin Zhao , Lin Guo , Chao Yang
中文翻译:

用邻碘代苯胺光诱导[3 + 2]烯烃环化:二氢吲哚的权宜之计
更新日期:2020-11-05
Synthesis ( IF 2.2 ) Pub Date : 2020-11-04 , DOI: 10.1055/s-0040-1705963 Wujiong Xia , Xinxin Zhao , Lin Guo , Chao Yang
|
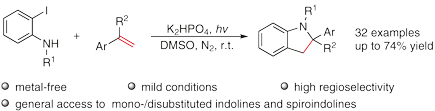
A highly regioselective [3+2] cyclization of alkenes with 2-iodoanilines under the irradiation of UV light is described. This general, metal-free strategy facilitates the direct preparation of 2-mono-/disubstituted indolines as well as spiroindolines through alkene carboamination in one step. Mechanistic studies suggested that the photochemical protocol proceeded via a radical pathway.
中文翻译:

用邻碘代苯胺光诱导[3 + 2]烯烃环化:二氢吲哚的权宜之计
描述了在紫外光照射下用2-碘苯胺对烯烃的高度区域选择性的[3 + 2]环化。这种一般的无金属策略有助于一步一步通过烯烃碳氨基化直接制备2-单-/二取代的二氢吲哚以及螺二氢吲哚。机理研究表明,光化学方案是通过自由基途径进行的。











































 京公网安备 11010802027423号
京公网安备 11010802027423号