当前位置:
X-MOL 学术
›
Adv. Synth. Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
A Metal‐ and Azide‐free Oxidative Coupling Reaction for the Synthesis of [1,2,3]Triazolo[1,5‐a]quinolines and their Application to Construct C−C and C‐P Bonds, 2‐Cyclopropylquinolines and Imidazo[1,5‐a]quinolines
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2020-10-29 , DOI: 10.1002/adsc.202001052 Zhi‐Hao Shang 1 , Zhen‐Xiao Zhang 1 , Wei‐Zhao Weng 1 , Yu‐Fei Wang 1 , Tian‐Wei Cheng 1 , Qiu‐Yi Zhang 1 , Li‐Qun Song 1 , Tian‐Qi Shao 1 , Kai‐Xuan Liu 1 , Yan‐Ping Zhu 1
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2020-10-29 , DOI: 10.1002/adsc.202001052 Zhi‐Hao Shang 1 , Zhen‐Xiao Zhang 1 , Wei‐Zhao Weng 1 , Yu‐Fei Wang 1 , Tian‐Wei Cheng 1 , Qiu‐Yi Zhang 1 , Li‐Qun Song 1 , Tian‐Qi Shao 1 , Kai‐Xuan Liu 1 , Yan‐Ping Zhu 1
Affiliation
|
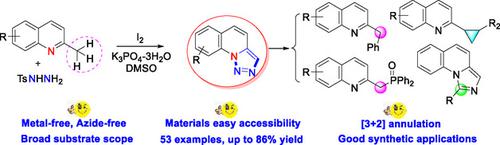
An iodine‐promoted one‐pot cascade oxidative annulation reaction has been developed for the synthesis of [1,2,3]triazolo[1,5‐a]quinolines from methyl azaarenes and N‐tosylhydrazines. The reaction has a broad substrate scope and can be easily scaled up to gram‐scale. 1,2,3‐Triazoles are an important skeletal structure for the construction of C−C and C−P bonds, 2‐cyclopropylquinolines and imidazo[1,5‐a]quinolines, for which different synthetic applications were explored.
中文翻译:

[1,2,3]三唑并[1,5-a]喹啉的无金属和叠氮化物氧化偶联反应及其在构建CC和CP键,2-环丙基喹啉和咪唑类中的应用[ 1,5‐a]喹啉
已经开发了一种碘促进的单锅级联氧化环化反应,用于从甲基氮杂芳烃和N-甲苯磺酰基肼合成[1,2,3]三唑并[1,5-α]喹啉。该反应具有广泛的底物范围,可以轻松放大至克级。1,2,3-三唑是构筑C-C和C-P键,2-环丙基喹啉和咪唑并[1,5-a]喹啉的重要骨架结构,已探索了它们的不同合成应用。
更新日期:2020-10-29
中文翻译:

[1,2,3]三唑并[1,5-a]喹啉的无金属和叠氮化物氧化偶联反应及其在构建CC和CP键,2-环丙基喹啉和咪唑类中的应用[ 1,5‐a]喹啉
已经开发了一种碘促进的单锅级联氧化环化反应,用于从甲基氮杂芳烃和N-甲苯磺酰基肼合成[1,2,3]三唑并[1,5-α]喹啉。该反应具有广泛的底物范围,可以轻松放大至克级。1,2,3-三唑是构筑C-C和C-P键,2-环丙基喹啉和咪唑并[1,5-a]喹啉的重要骨架结构,已探索了它们的不同合成应用。









































 京公网安备 11010802027423号
京公网安备 11010802027423号