当前位置:
X-MOL 学术
›
Chin. J. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Fe‐Catalyzed Pictet‐Spengler‐Type Cyclization via Selective Four‐Electron Reductive Functionalization of CO2
Chinese Journal of Chemistry ( IF 5.5 ) Pub Date : 2020-10-18 , DOI: 10.1002/cjoc.202000521 Wen‐Duo Li 1, 2 , Jie Chen 1 , Dao‐Yong Zhu 1 , Ji‐Bao Xia 1, 2
Chinese Journal of Chemistry ( IF 5.5 ) Pub Date : 2020-10-18 , DOI: 10.1002/cjoc.202000521 Wen‐Duo Li 1, 2 , Jie Chen 1 , Dao‐Yong Zhu 1 , Ji‐Bao Xia 1, 2
Affiliation
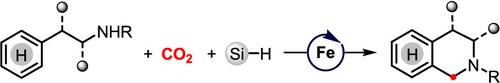
|
Herein, we describe a novel catalytic Pictet‐Spengler‐type cyclization using CO2 as a nontoxic and sustainable C1 feedstock with environmentally benign and non‐precious‐metal iron as catalyst. The reaction is achieved by selective four‐electron reduction of CO2 into methylene level intermediate through carefully tuning the reaction parameters. A variety of tetrahydro‐β‐carbolines and other nitrogen‐containing heterocycles can be easily obtained under mild conditions. Mechanistic studies have shown that tetrahydro‐β‐carbolines are probably obtained via spiroindolenine intermediates.
中文翻译:

铁通过选择性四电子还原功能化CO催化的Pictet-Spengler型环化
在本文中,我们描述了一种新颖的Pictet-Spengler型催化式环化反应,该反应使用CO 2作为无毒且可持续的C1原料,对环境无害且非贵金属铁作为催化剂。通过仔细调节反应参数,将CO 2选择性四电子还原为亚甲基级中间体,可以实现反应。在温和的条件下,很容易获得各种四氢-β-咔啉和其他含氮杂环。机理研究表明,四氢-β-咔啉可能是通过螺环吲哚烯中间体获得的。
更新日期:2020-10-18

中文翻译:

铁通过选择性四电子还原功能化CO催化的Pictet-Spengler型环化
在本文中,我们描述了一种新颖的Pictet-Spengler型催化式环化反应,该反应使用CO 2作为无毒且可持续的C1原料,对环境无害且非贵金属铁作为催化剂。通过仔细调节反应参数,将CO 2选择性四电子还原为亚甲基级中间体,可以实现反应。在温和的条件下,很容易获得各种四氢-β-咔啉和其他含氮杂环。机理研究表明,四氢-β-咔啉可能是通过螺环吲哚烯中间体获得的。












































 京公网安备 11010802027423号
京公网安备 11010802027423号