Journal of Environmental Chemical Engineering ( IF 7.4 ) Pub Date : 2020-10-19 , DOI: 10.1016/j.jece.2020.104601 Angie E. Orduz , Carolina Acebal , Graciela Zanini
|
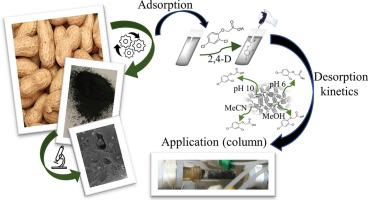
The desorption kinetic of 2,4-Dichlorophenoxyacetic acid (2,4-D) herbicide initially adsorbed on activated carbon from peanut shells (PSAC) was studied. The herbicide was adsorbed at pH 3.0 and desorbed in aqueous solutions at pH 6.0 and 10.0, in acetonitrile (MeCN) and methanol (MeOH). The determined desorption rate constants ) were 0.362 s−1, 0.530 s−1, 0.638 s−1, and 0.876 s−1, respectively. The activation energy of desorption (Ea) was determined as 7.4 kJ/mol at pH 6.0 and 11.5 kJ/mol at pH 10.0 using the Arrhenius equation. Adsorption isotherms in aqueous solution at different pH and different solvents showed that the affinity of 2,4-D on the surface of PSAC is higher in aqueous solvents than in organic solvents. PSAC was successfully applied as a low-cost carbonaceous material to be packed in columns for analytical purposes as solid-phase extraction procedure (SPE). The results obtained allowed MeOH to be chosen as the right solvent for this analytical application. In addition, the knowledge of this process could be extremely helpful to avoid a new solid waste after retaining the contaminants in water purification processes and even recovering the carbonaceous material and the solvent in a simple way. The desorption kinetic of 2,4-D in several solvents including water a different pH and 2,4-D adsorption isotherms in organic solvents have not been reported so far.
中文翻译:

花生壳中的活性炭:2,4-D解吸动力学研究,用作绿色材料用于分析目的
研究了最初吸附在花生壳(PSAC)活性炭上的2,4-二氯苯氧基乙酸(2,4-D)除草剂的解吸动力学。将除草剂在pH 3.0下吸附,并在pH 6.0和10.0的水溶液中,在乙腈(MeCN)和甲醇(MeOH)中解吸。确定的解吸速率常数)为0.362小号-1,0.530小号-1,0.638小号-1,和0.876小号-1,分别。解吸的活化能(E a使用Arrhenius方程确定pH为6.0时为7.4 kJ / mol,pH 10.0为11.5 kJ / mol。在不同pH和不同溶剂下的水溶液中的吸附等温线表明,PSAC表面上2,4-D的亲和力在水性溶剂中比在有机溶剂中更高。PSAC已成功地用作低成本的碳质材料,并以固相萃取程序(SPE)的形式用于色谱分析。获得的结果允许选择MeOH作为该分析应用的正确溶剂。另外,该方法的知识对于避免在水净化过程中保留污染物后甚至避免以简单方式回收含碳物质和溶剂后产生的新固体废物,将非常有帮助。2的解吸动力学











































 京公网安备 11010802027423号
京公网安备 11010802027423号