Acta Biomaterialia ( IF 9.7 ) Pub Date : 2020-10-13 , DOI: 10.1016/j.actbio.2020.09.052 W Benton Swanson 1 , Zhen Zhang 1 , Kemao Xiu 1 , Ting Gong 1 , Miranda Eberle 2 , Ziqi Wang 3 , Peter X Ma 4
|
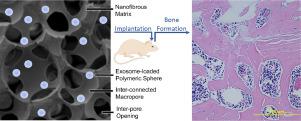
Biomimetic bone regeneration methods which demonstrate both clinical and manufacturing feasibility, as alternatives to autogenic or allogenic bone grafting, remain a challenge to the field of tissue engineering. Here, we report the pro-osteogenic capacity of exosomes derived from human dental pulp stem cells (hDPSCs) to facilitate bone marrow stromal cell (BMSC) differentiation and mineralization. To support their delivery, we engineered a biodegradable polymer delivery platform to improve the encapsulation and the controlled release of exosomes on a tunable time scale from poly(lactic-co-glycolic acid) (PLGA) and poly(ethylene glycol) (PEG) triblock copolymer microspheres. Our delivery platform integrates within three-dimensional tissue engineering scaffolds to enable a straightforward surgical insertion into a mouse calvarial defect. We demonstrate the osteogenic potential of these functional constructs in vitro and in vivo. Controlled release of osteogenic hDPSC-derived exosomes facilitates osteogenic differentiation of BMSCs, leading to mineralization to a degree which is comparable to exogenous administration of the same exosomes in human and mouse BMSCs. By recruiting endogenous cells to the defects and facilitating their differentiation, the controlled release of osteogenic exosomes from a tissue engineering scaffold demonstrates accelerated bone healing in vivo at 8 weeks. Exosomes recapitulate the advantageous properties of mesenchymal stem/progenitor cells, without manufacturing or immunogenic concerns associated with transplantation of exogenous cells. This biomaterial platform enables exosome-mediated bone regeneration in an efficacious and clinically relevant way.
中文翻译:

控制释放促矿化外泌体的支架,无需细胞移植即可促进颅面骨愈合
仿生骨再生方法作为自体或同种异体骨移植的替代方案,证明了临床和制造的可行性,仍然是组织工程领域的一个挑战。在这里,我们报道了源自人牙髓干细胞(hDPSC)的外泌体的促成骨能力,以促进骨髓基质细胞(BMSC)分化和矿化。为了支持它们的递送,我们设计了一个可生物降解的聚合物递送平台,以改善外泌体的封装和在可调时间尺度上由聚乳酸-乙醇酸共聚物(PLGA)和聚乙二醇(PEG)三嵌段的控制释放共聚物微球。我们的输送平台集成在三维组织工程支架内,可以通过手术直接插入小鼠颅骨缺损中。我们在体外和体内证明了这些功能结构的成骨潜力。成骨 hDPSC 衍生的外泌体的受控释放促进 BMSC 的成骨分化,导致矿化程度与在人和小鼠 BMSC 中外源施用相同外泌体相当。通过将内源细胞募集到缺损处并促进其分化,从组织工程支架中受控释放成骨外泌体,证明在 8 周时可加速体内骨愈合。外泌体概括了间充质干细胞/祖细胞的有利特性,而没有与外源细胞移植相关的制造或免疫原性问题。该生物材料平台能够以有效且临床相关的方式实现外泌体介导的骨再生。



























 京公网安备 11010802027423号
京公网安备 11010802027423号