Current Analytical Chemistry ( IF 1.7 ) Pub Date : 2020-10-31 , DOI: 10.2174/1573411015666191114143128 Şerife Parlayıcı 1 , Kübra Tuna Sezer 1 , Erol Pehlivan 1
|
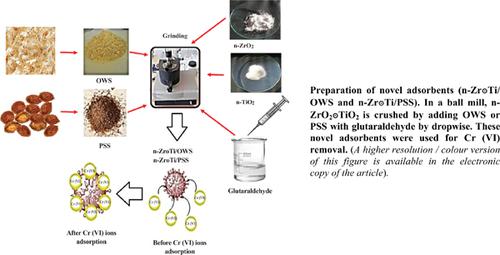
Background: In this work, Cr (VI) adsorption on nano-ZrO2๏TiO2 impregnated orange wood sawdust (Zr๏Ti/OWS) and nano-ZrO2๏TiO2 impregnated peach stone shell (Zr๏Ti/PSS) was investigated by applying different adsorption parameters such as Cr (VI) concentrations, contact time, adsorbent dose, and pH for all adsorbents.
Methods: The adsorbents were characterized by SEM and FT-IR. The equilibrium status was achieved after 120 min of contact time and optimum pH value around 2 were determined for Cr (VI) adsorption. Adsorption data in the equilibrium is well-assembled by the Langmuir model during the adsorption process.
Results: Langmuir isotherm model showed a maximum adsorption value of OWS: 21.65 mg/g and Zr๏Ti/OWS: 27.25 mg/g. The same isotherm displayed a maximum adsorption value of PSS: 17.64 mg/g, and Zr๏Ti/PSS: 31.15 mg/g. Pseudo-second-order kinetic models (R2=0.99) were found to be the best models for describing the Cr (VI) adsorption reactions.
Conclusion: Thermodynamic parameters such as changes in ΔG°, ΔH°, and ΔS° have been estimated, and the process was found to be spontaneous.
中文翻译:

纳米ZrO 2 / TiO 2浸渍的橙色木屑和桃石壳吸附剂用于去除Cr(VI)
背景:在这项工作中,研究了Cr(VI)在纳米ZrO2๏TiO2浸渍的橙色木屑(Zr๏Ti / OWS)和纳米ZrO2๏TiO2浸渍的桃石外壳(Zr๏Ti / PSS)上的吸附。吸附参数,例如所有吸附剂的Cr(VI)浓度,接触时间,吸附剂剂量和pH。
方法:用SEM和FT-IR对吸附剂进行表征。在120分钟的接触时间后达到了平衡状态,并确定了Cr(VI)吸附的最佳pH值约为2。在吸附过程中,朗缪尔模型很好地平衡了平衡中的吸附数据。
结果:Langmuir等温线模型显示OWS的最大吸附值为:21.65 mg / g,Zr๏Ti / OWS的最大吸附值为:27.25 mg / g。相同的等温线显示PSS的最大吸附值:17.64 mg / g,Zr 3 Ti / PSS:31.15 mg / g。伪二级动力学模型(R2 = 0.99)被发现是描述Cr(VI)吸附反应的最佳模型。
结论:已经估算出热力学参数,例如ΔG°,ΔH°和ΔS°的变化,并且发现该过程是自发的。











































 京公网安备 11010802027423号
京公网安备 11010802027423号