当前位置:
X-MOL 学术
›
ChemBioChem
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Covalent Linkage and Macrocylization Preserve and Enhance Synergistic Interactions in Catalytic Amyloids
ChemBioChem ( IF 2.6 ) Pub Date : 2020-09-21 , DOI: 10.1002/cbic.202000645 Zsofia Lengyel-Zhand 1 , Liam R Marshall 1 , Maximilian Jung 2 , Megha Jayachandran 1 , Min-Chul Kim 1 , Austin Kriews 1 , Olga V Makhlynets 1 , H Christopher Fry 3 , Armin Geyer 2 , Ivan V Korendovych 1
ChemBioChem ( IF 2.6 ) Pub Date : 2020-09-21 , DOI: 10.1002/cbic.202000645 Zsofia Lengyel-Zhand 1 , Liam R Marshall 1 , Maximilian Jung 2 , Megha Jayachandran 1 , Min-Chul Kim 1 , Austin Kriews 1 , Olga V Makhlynets 1 , H Christopher Fry 3 , Armin Geyer 2 , Ivan V Korendovych 1
Affiliation
|
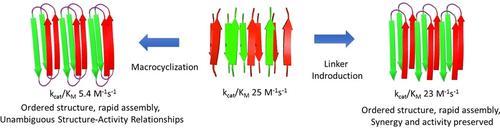
How might complexity arise from short peptides? Synergistic interactions between the side chains of catalytic amyloids are locked into place by linkers between the peptides, and these new designs result in faster assembly and asymmetric assemblies. These could serve as a potential link in the evolutionary path between abiotic peptides and complex enzymes.

中文翻译:

共价连接和大环化保持并增强催化淀粉样蛋白的协同相互作用
短肽如何产生复杂性?催化淀粉样蛋白侧链之间的协同相互作用通过肽之间的连接体锁定到位,这些新设计导致更快的组装和不对称组装。这些可以作为非生物肽和复杂酶之间进化路径的潜在联系。

更新日期:2020-09-21

中文翻译:

共价连接和大环化保持并增强催化淀粉样蛋白的协同相互作用
短肽如何产生复杂性?催化淀粉样蛋白侧链之间的协同相互作用通过肽之间的连接体锁定到位,这些新设计导致更快的组装和不对称组装。这些可以作为非生物肽和复杂酶之间进化路径的潜在联系。












































 京公网安备 11010802027423号
京公网安备 11010802027423号